Original Article
A Comprehensive Description of Naturopathic Care for Advanced Cancers: Outcomes from the Canadian/US Integrative Oncology Study
Dugald Seely,1,2 ND, MSc, Mark Legacy,1,2 CCRP, Ellen Conte,2 ND, Erica Rizzolo,1,2 ND, Erin Sweet,3 ND, MPH, Peih F. Chiang,3 ND, Linda Dale,3 CCRP, Athanasios Psihogios,1 MPH, Julie Ennis,1 PhD, Gurdev Parmar,4 ND, Eric Marsden,5 ND, Michael Reid,6 ND, Dan Rubin,7 ND, Paul Reilly,8 ND, LAc, Michael Traub,9 ND, Eleonora Naydis,10 ND, LAc, Tim Ramsay,11 PhD, and Leanna J. Standish,3 ND, PhD, LAc
ABSTRACT
Background: There is a paucity of real-world data on the treatments naturopathic doctors (NDs) use for supportive cancer care. We conducted an observational cohort study to comprehensively describe the treatments NDs with experience in cancer care recommend to their patients.
Methods: Patients with advanced breast, colorectal, ovarian, or pancreatic cancer were recruited from 12 North American naturopathic clinics and followed for 2 to 3 years. Therapeutic recommendations were abstracted from clinic records.
Results: 384 participants (154 breast, 112 colorectal, 71 ovarian, 47 pancreatic) were included in the analyses. The median number of ND visits was 5. The most common types of recommendations were natural health products (NHPs, 99% of participants), nutrition guidance (88%), and parenteral therapies (81%). Mental health (33%) and Traditional Chinese Medicine (29%) were least common. Participants were recommended a median of 11 NHPs throughout the study, with 430 unique products recommended across all participants. Nutrition guidance heavily favoured encouraging foods rather than discouraging them (83% vs. 17% of all dietary recommendations, respectively). Vitamin D, curcumin, intravenous vitamin C, increasing protein intake, and exercise were recommended to at least 50% of participants across each cancer type. Other common recommendations included melatonin, fish oil, Trametes versicolor, subcutaneous mistletoe, increasing vegetable intake, and eating behaviour changes. Recommendations were fairly uniform between cancer types, with the most variation seen in NHPs.
Conclusion: NDs frequently recommend NHPs, nutrition guidance, and parenteral therapies for people with advanced cancer. The diversity of unique recommendations suggests individualized care, yet some commonly used treatments demonstrate a degree of consistency.
Key Words Naturopathic medicine, integrative medicine, integrative oncology, naturopathic cancer care, supportive cancer care, naturopathic oncology, naturopathic treatments, treatment recommendations, naturopathic doctor, naturopathic physician, breast cancer, ovarian cancer, colorectal cancer, pancreatic cancer
INTRODUCTION
Traditional, complementary and integrative medicine (TCIM) is reported to be used by 40% to 80% of people with cancer.1–3 Under the TCIM umbrella, naturopathic medicine (NM) is a system of care provided by naturopathic doctors (NDs) that includes the use of nutritional guidance, dietary supplements, herbal medicine, homeopathic remedies, intravenous and injection therapy, acupuncture, counselling and emotional support, and various lifestyle practices.4 Naturopathic medicine is one of the most commonly practiced systems of TCIM in the Western world,4 with approximately 8,000 licensed NDs in North America.5 Studies suggest NM use is higher among those with cancer than the general population,6 although the exact prevalence of use is unknown.
The goals of NM in cancer care include educating patients on adopting healthy lifestyles, managing side effects, improving treatment response, reducing recurrence risk, and optimizing overall health.7 Although specialization within naturopathic medicine is not currently recognized by most state and provincial regulators, fellowship certification is established for cancer care through the American Board of Naturopathic Oncology, which is part of the Oncology Association of Naturopathic Physicians (OncANP).8 Currently, 133 NDs across North America are Fellows of the American Board of Naturopathic Oncology, and about 400 NDs are members of the OncANP (email communication with OncANP Executive Director, Corey Murphy, March 6, 2025).
To date, clinical practice guidelines, which establish standard of care, are largely absent from NM. Limited guidelines for the broader field of integrative oncology have been published in recent years9,10 but are not specific to naturopathic practice nor comprehensive enough to guide complex care. In 2019, the OncANP published a Principles of Care guidance document, which describes the naturopathic medical approach to cancer care.7 The document provides guidance on how to deliver patient-centred care from diagnosis through to survivorship; however, it does not discuss treatments. The lack of standardization leads to limited knowledge of what recommendations NDs make for individuals with cancer. This is particularly relevant given the broad range of therapies available to NDs. To our knowledge, Standish et al. has published the only study reporting on naturopathic oncology care, which included patients with breast cancer of any stage.11 Several surveys of NDs have also explored management for cancer;12–14 however, survey responses may not accurately reflect real-world practice, and some data are likely outdated. Despite these contributions, gaps remain. Describing how NDs care for those with cancer enhances our understanding of the field, helps identify areas for improvement, improves transparency, and may aid the development of clinical practice guidelines and standards of care.
The Canadian/US Integrative Oncology Study is an observational cohort study with the overarching goal of studying naturopathic oncology in a real-world setting. The primary objective of the project was to measure survival in patients with advanced stage breast, colorectal, ovarian, or pancreatic cancer who consulted with an ND. Secondary objectives included describing the scope of treatments recommended to patients by NDs, comparing survival amongst enrolled participants based on the number of ND visits and the frequency of use of verifiably received naturopathic treatments, collecting health-related quality of life (QOL) data, estimating costs of cancer care, and gathering information on participants’ qualitative experience of care.
In this paper, we describe the scope of therapeutic recommendations made by NDs to people with advanced breast, colorectal, ovarian, or pancreatic cancer. Survival, QOL, cost, and data on qualitative experiences will be reported in separate publications.
METHODS
This paper was written alongside our reports on survival, cost, QOL, and qualitative experiences. The study methods are similar across these publications; thus, there will be significant overlap.
Study Design
This was an observational study of patients with advanced stage breast, colorectal, ovarian, or pancreatic cancer who consulted with an ND at one of 12 clinics in North America. Recruitment and follow-up were prospective. Clinics were instructed to approach all eligible patients; however, we did not have a method to verify consecutive recruitment.
Setting
The study was conducted at 12 outpatient naturopathic clinics located in Canada (n = 5) and the US (n = 7). The primary coordinating centres were the Bastyr Center for Natural Health in Seattle, Washington, and The Centre for Health Innovation in Ottawa, Ontario. Additional sites were: Integrated Health Clinic Cancer Care Centre, Fort Langley, British Columbia; Marsden Centre for Excellence in Integrative Medicine, Vaughan, Ontario; Health Source Integrative Medical Centre, Kitchener, Ontario; Vital Victoria Naturopathic Clinic, Victoria, British Columbia; Naturopathic Specialists LLC, Scottsdale, Arizona; Salish Care Center, Fife, Washington; Hawaii Integrative Oncology, Kailua-Kona, Hawaii; Tree of Health Integrative Medicine, Woodinville, Washington; Seattle Integrative Cancer Center, Seattle, Washington; Advanced Integrative Medical Science Institute, Seattle, Washington. Clinics were selected for their focus in cancer care and NDs with a depth of experience in the field. A total of 29 NDs provided care to at least one study participant. All NDs had experience in a cancer care setting, and 15 were Fellows of the American Board of Naturopathic Oncology.
Participants
Eligible participants were adults 18 years of age and over who presented for care to a participating clinic with a confirmed diagnosis of one of the following:
- ▪ Metastatic breast cancer
(de novo stage IV or recurrent)
- ▪ Metastatic colorectal cancer
(de novo stage IV or recurrent)
- ▪ Advanced ovarian cancer
(stage III or IV, de novo or recurrent)
- ▪ Advanced pancreatic cancer
(stage III or IV, de novo or recurrent)
Stages were determined per the American Joint Committee on Cancer’s staging guidelines (version 7). Participants additionally had to be citizens or permanent residents of the country in which they were receiving treatment. All participants signed an informed consent form in writing prior to participating. Recruitment began in June 2015 and concluded in March 2020. Participants were followed for at least 2 years and up to 3 years. Variable follow-up time occurred due to funding restraints, which arose due to a longer than anticipated recruitment period. All participants were offered a $200 stipend upon enrolment to be used towards any practitioner consultation at the participant’s naturopathic clinic.
Regulatory Adherence
Ethics approval was obtained from the Research Ethics Board of the Canadian College of Naturopathic Medicine and the Institutional Review Board at the Office of Research Integrity at Bastyr University prior to study initiation. The study was registered with clinicaltrials.gov, NCT02494037.
Outcomes
The primary study outcome was to provide a comprehensive description of the type and frequency of naturopathic recommendations made to patients with advanced cancer. Only the incidence and frequency of these recommendations were recorded; compliance was not assessed.
Data Sources
Data were collected from naturopathic clinic records and hospital medical records. Naturopathic clinic records were reviewed from the first visit until the study end date, and conventional records were reviewed from diagnosis until the study end date.
Sample Size
The total expected study sample size was 400 patients, of which it was estimated 150 would have breast cancer, 150 would have colorectal cancer, 50 would have ovarian cancer, and 50 would have pancreatic cancer. The sample size was determined based on pragmatic considerations related to the number of estimated eligible patients the clinics would see, the expected rate of enrollment, study duration, and available funding.
Quantitative Variables
At each visit, all recommendations made by NDs were abstracted and separated into distinct categories using the Research Electronic Data Capture (REDCap) platform. Entries were verified by a second team member. Each recommendation was then placed into one of the following categories: natural health products (NHPs), nutrition, parenteral therapies, prescription and over-the-counter (OTC) medications, hyperthermia, Traditional Chinese Medicine (TCM) practices (e.g., acupuncture, moxibustion), physical medicine and body-based therapies (e.g., exercise, massage therapy, hydrotherapy), and mental health.
Nutrition recommendations were divided into encouragements and discouragements. Dietary encouragements encompassed recommendations to start, continue, or increase a specific food, type of food, or eating behaviour. Discouragements included recommendations to decrease, hold, or stop these items. A recommendation for a prescription or OTC medication included recommending an OTC medication (e.g., ibuprofen), writing a prescription for a medication (for practitioners with prescribing authority), or advising the patient to speak with a medical doctor or nurse practitioner about a medication. Cannabis was abstracted in the same manner given it is not considered an NHP in Canada. Hyperthermia recommendations encompassed both whole body and locoregional hyperthermia (LRHT). A recommendation was categorized as mental health support if it could conceivably help improve the participant’s mental health and did not fit in any other recommendation category (e.g., recommendations to see a therapist, or to practice meditation).
In a companion paper reporting on the survival of our participants,15 we included an analysis on survival time based on the number of intravenous vitamin C (IVC), IV mistletoe, and hyperthermia treatments. For this analysis, we collected the number of verifiably received IV and hyperthermia treatments. In this paper, we additionally report on the proportion of participants who started IV therapies and hyperthermia compared with those who were recommended those treatments.
Statistical Methods
Results were analyzed using frequency distributions and descriptive statistics. Standard deviation (SD) was used as a method of dispersion for mean values. Inter-quartile range (IQR) or range was used as a method of dispersion for median values. Items were included in the results if they were recommended to at least 10% of participants in at least one cancer type. The Chi-squared test and standardized residuals were used to analyze differences in recommendations between cancer types. Cramer’s V was used to determine the strength of the association. Values ≤ 0.3 were considered weak, values between 0.3 and 0.5 were considered moderate, and values > 0.5 were considered strong. Standardized residuals above |2.00| were considered significant. A significant positive residual (i.e., ≥ 2.00) indicates that a therapy or treatment was recommended more often than expected in that cancer type if it is assumed there is no difference between cancer types. A significant negative residual (i.e., ≤ −2.00) indicates a recommendation was made less often than expected. All results from the statistical analyses are presented in Appendix A, Tables A1–A10.
Data Availability Statement
Datasets used in this study are available by request only. Please contact Dugald Seely, dseely@thechi.ca, if you would like to request access to any datasets.
RESULTS
Participant Characteristics
In total, 400 patients were enrolled. Sixteen were deemed screen failures (i.e., ineligible after being enrolled), leaving 384 participants included in the analyses. One hundred fifty-four had breast cancer, 112 had colorectal cancer, 71 had ovarian cancer, and 47 had pancreatic cancer. Figure 1 shows participant flow throughout the study.
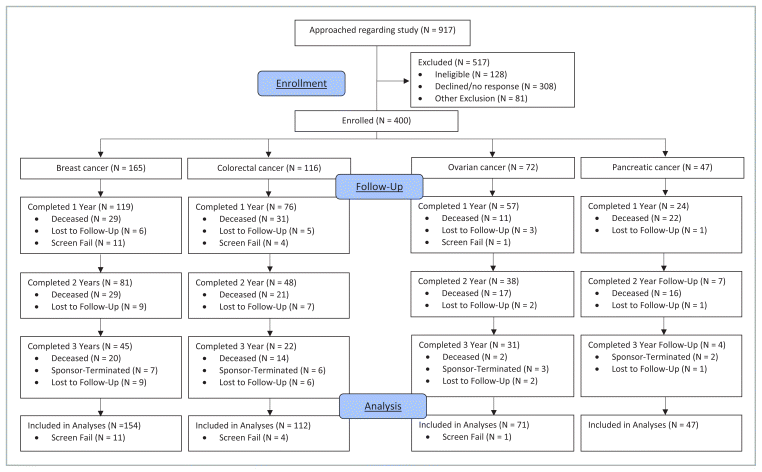
FIGURE 1 Participant Flow Diagram. Patients were only recorded as approached if an investigator believed they were eligible for the study and if the patient was interested in participating. Screen fail: patient was enrolled in the study but was later found to be ineligible.
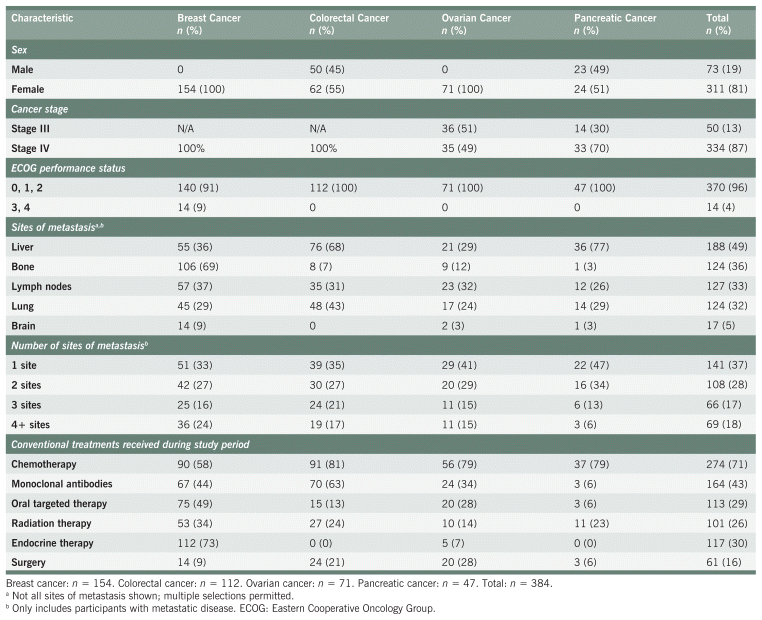
Mean age at enrolment was 57.3 ± 11.8 years. Fifty participants (36 ovarian, 14 pancreatic) had stage III disease upon enrolment; all other participants had metastatic disease. Of those with metastatic disease, 142 (43%) were diagnosed de novo. Eighty-eight percent of participants received conventional cancer treatments before the study period and 90% received treatments during the study period. Only 4 participants did not receive any conventional treatments. Table 1 shows participants’ baseline clinical status and demographic characteristics separated by cancer type.
TABLE 1 Clinical Status and Demographics
Naturopathic Consultations
The median number of ND visits across the study was 5 (IQR 3–8). This differed slightly by cancer type:
- ▪ Breast: median 6 (IQR 3–9)
- ▪ Colorectal: median 4 (IQR 2–7.5)
- ▪ Ovarian: median 5 (IQR 3–8)
- ▪ Pancreatic: median 4 (IQR 2–7)
Over time, participants across all cancer types had fewer ND consultations. For those who were still alive, 50% had a consultation in their second year on study and 43% had a consultation in their third year. For those who continued to see their ND, the number of visits also declined. The mean number of ND visits was 4.7 ± 3.2 in the first year, 2.9 ± 2.1 in the second year, and 2.4 ± 1.9 in the third year. Thirty-eight participants (10%) had only a single visit with their ND.
Recommendations – Overview
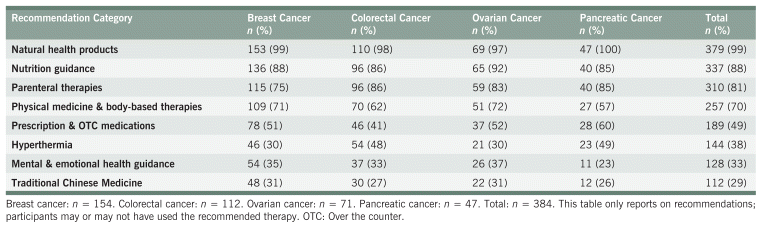
Natural health products, nutrition, and parenteral therapies were the most common treatment modalities, recommended to 99%, 88%, and 81% of all participants, respectively. Recommendations for mental health and TCM were made least often (33% and 29%, respectively). In general, the proportion of recommendations between cancer types was within 10% of others within the collective cohort. Notable exceptions include a higher incidence of hyperthermia recommendations in colorectal and pancreatic cancer, a higher incidence of prescription and OTC medications in pancreatic cancer, and a lower incidence of mental health and physical medicine recommendations in pancreatic cancer. Table 2 shows how frequently all modalities of naturopathic treatments were recommended to participants, separated by cancer type.
TABLE 2 Recommendations by Treatment Modality
Recommendations – Natural Health Products
In total, there were 430 unique NHP recommendations across all participants. The median number of distinct recommendations per participant throughout the study was 11 (IQR 7–15). Approximately half (median = 6) of these recommendations were made at the initial consultation. Between cancer types, those with pancreatic cancer were recommended fewer NHPs (median = 7) compared with the other cancer types (breast: 12, colorectal: 10, ovarian: 12). There was a moderate positive correlation between the number of NHP recommendations and number of ND visits (r = 0.57, p < 0.001).
Table 3 shows the frequency of different categories of NHP recommendations. The most common items were vitamins and minerals (recommended to 93% of all participants), herbal medicine (83%), and hormone-based products (e.g., melatonin) (68%). Homeopathic remedies (18%), fibre supplements (6%), and essential oils (4%) were the least common.
TABLE 3 Natural Health Product Recommendations by Treatment Category
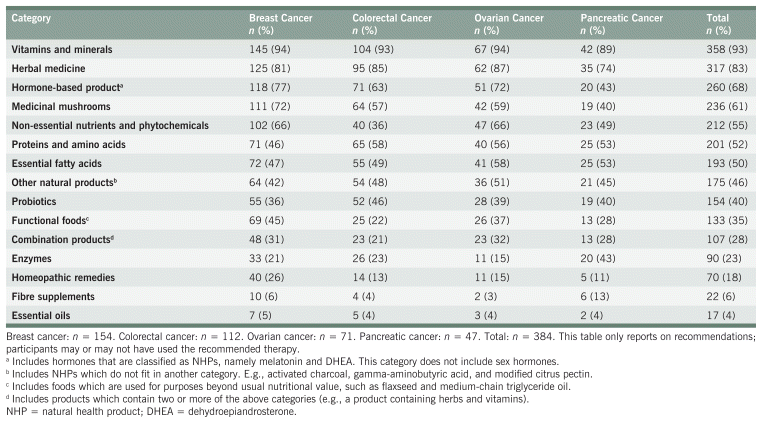
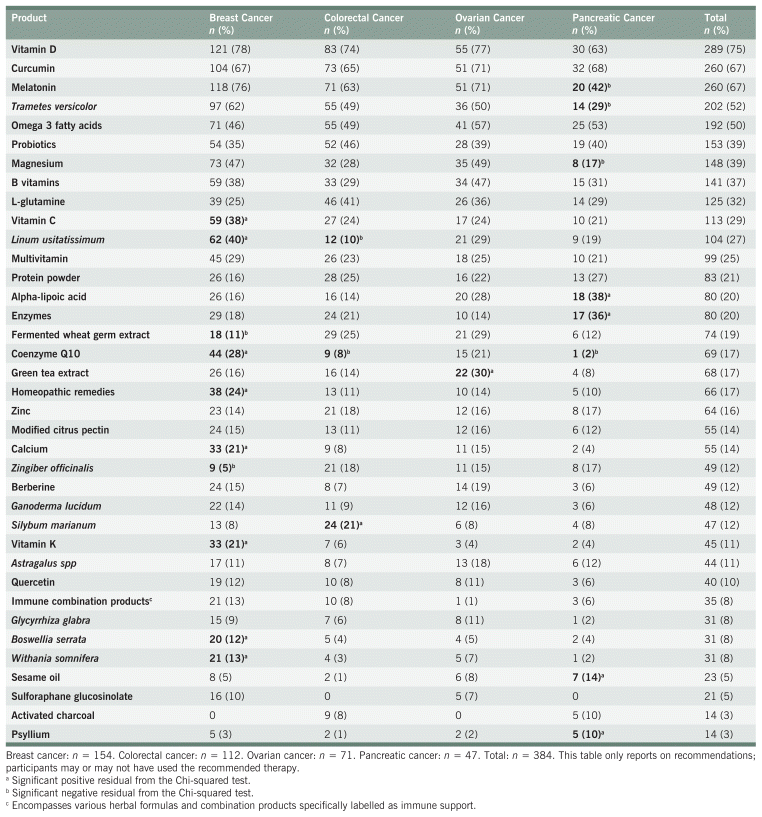
Table 4 shows the frequency of specific NHP recommendations. The most common was vitamin D, made to 75% of participants. Only vitamin D and curcumin were recommended to at least 50% of participants across all cancer types. Other top recommendations included melatonin, Trametes versicolor, and omega-3 fatty acids, each recommended to at least 50% of the total population. A statistically significant Chi-squared test statistic was seen for 20 NHPs, indicating the distribution of recommendations was not uniform across all cancer types; however, all associations were weak (Cramer’s V < 0.3). Several NHPs also showed statistically significant associations with one cancer type (i.e., a positive or negative residual). The breast cancer cohort had the most positive associations (n = 9 NHPs), indicating many treatments were recommended more frequently than expected for those with breast cancer. The pancreatic cancer cohort had the most negative associations (n = 4), indicating many treatments were recommended less frequently than expected for these participants.
TABLE 4 Natural Health Product Recommendations by Specific Product
Recommendations – Nutrition
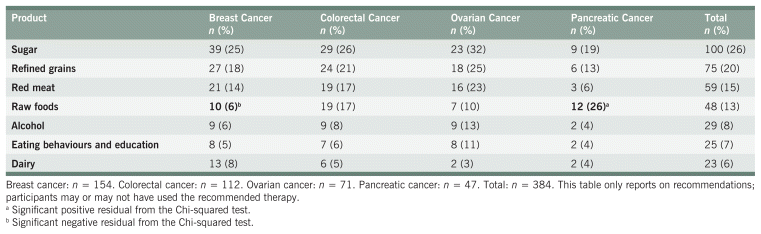
Tables 5 and 6 show the most common nutrition encouragements and discouragements, respectively. Dietary encouragements were made much more often than discouragements (83% vs. 17% of all dietary recommendations, respectively). This trend was consistent across each cancer type. The top ten most encouraged dietary items were: protein-rich foods (e.g., poultry, fish, legumes), vegetables, eating behaviour changes and nutrition education, fruits, tea, nuts and seeds, fats, fasting (e.g., before chemotherapy, intermittent fasting), water, and whole grains. Protein-rich foods was the only recommendation made to at least 50% of participants across all cancer types. Tea recommendations were primarily green tea (63% of all tea recommendations) and herbal teas (36%) The most common eating behaviour recommendations were increasing caloric intake (23% of all recommendations) and eating small frequent meals (20%). The only items that were mainly discouraged were sugar, refined grains, red meat, raw foods, and alcohol. One exception is in participants with pancreatic cancer, where red meat was encouraged slightly more often than it was discouraged (8% versus 6%, respectively). Teas (encouragement) and raw foods (discouragement) were the only items with significant non-uniformity across cancer types. Raw foods were discouraged more often than expected in participants with pancreatic cancer and less often than expected in participants with breast cancer.
TABLE 5 Dietary Encouragements
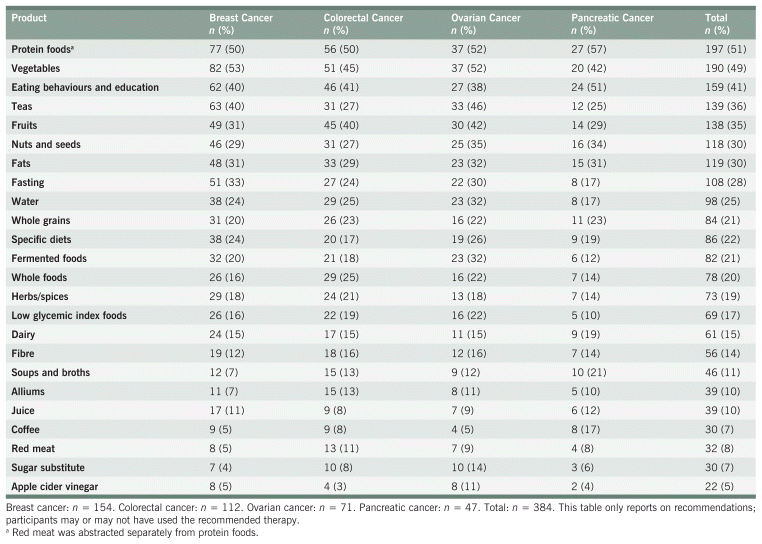
TABLE 6 Dietary Discouragements
Parenteral Therapies
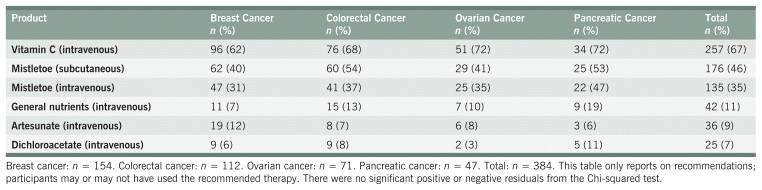
Table 7 shows the most common parenteral therapies recommended to participants. Only 6 parenteral therapies were recommended to ≥ 10% of participants in at least one cancer type. These included IVC, IV and subcutaneous (SC) mistletoe, general nutrient infusions, and infusions of artesunate and dichloroacetate (DCA). IVC was by far the most common, recommended to at least 60% of participants across cancer types. Subcutaneous mistletoe was recommended to at least 40% of participants, and IV mistletoe was recommended to at least 30% of participants across cancer types. Dichloroacetate and artesunate were least common, both recommended to ≤ 10% of participants in total. No parenteral therapies showed significant non-uniformity between cancer types based on the Chi-squared test.
TABLE 7 Parenteral Therapy Recommendations
Other Notable Recommendations
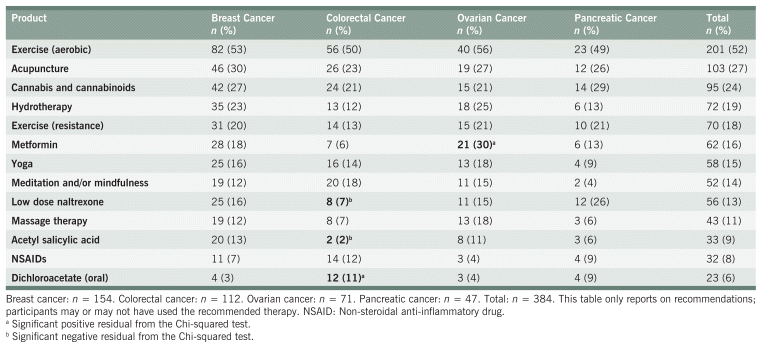
Exercise was recommended to 57% of participants, with aerobic exercise (52%) being far more common than resistance training (18%). Cannabis and cannabinoids were recommended to 24% of participants. Hydrotherapy and massage therapy were recommended to 19% and 11% of participants, respectively. Metformin (16%) and low-dose naltrexone (15%) were the most recommended prescription medications. Yoga and meditation/mindfulness were the most recommended activities to support mental health, each made to approximately 15% of participants. Acupuncture was recommended to 27% of participants. All prescription and OTC medications (except non-steroidal anti-inflammatory drugs), as well as hydrotherapy recommendations, were significantly different between cancer types per the Chi-squared test; however, associations were weak. Table 8 fully describes these recommendations.
TABLE 8 Other Common Recommendations
Treatments Received
Of the 144 participants who were recommended hyperthermia, 96 (67%) received treatments at one of the study clinics. Compared with the number of participants who were recommended IV therapies, 69% received one or more IVC infusions, 63% received IV mistletoe, 38% received general nutrient infusions, 14% received IV artesunate, and 4% received IV DCA.
DISCUSSION
In this multicentre North American observational study, we comprehensively described naturopathic treatment recommendations for patients with late-stage breast, colorectal, ovarian, and pancreatic cancer. Participants were commonly recommended NHPs, IVC, and aerobic exercise, and received dietary counselling. Standish et al. previously provided a summary of naturopathic treatment recommendations for patients with any stage breast cancer.11 Some data are consistent with our breast cancer cohort, including the high frequency of recommendations for vitamin D, curcumin, omega 3 fatty acids, and Trametes versicolor. Notably, dietary and exercise recommendations are much higher in both our breast cancer cohort and the entire study population. The reasons for these discrepancies are not clear. IV therapy recommendations were also significantly higher in our study; however, this is likely because only 12% of participants from Standish et al. had metastatic disease, which is where IVC has been most heavily studied.16
Our results demonstrate that naturopathic care for advanced cancer has some degree of consistency in the most common treatment recommendations while also offering a wide range of treatments. The consistency is demonstrated by the frequency with which certain NHPs, nutrition, and exercise interventions were recommended; vitamin D, curcumin, IVC, and aerobic exercise were all recommended to at least 50% of participants across all cancer types, with several other recommendations above 35%. This is notable given the large number of clinics involved in this study and the lack of practice guidelines in naturopathic cancer care. The individualized nature of care is evidenced by the wide array of unique treatment recommendations, with many having a low overall frequency of use. One possible explanation is the lack of practice guidelines which, given the large number of practitioners and locations of practice, could yield a variety of different treatments being used for similar conditions. However, this individuality is in line with principles of naturopathic medicine, namely, to treat the whole person and to identify the root cause of illness.4 These principles emphasize a holistic and individualized approach to care that likely explains the broad scope of therapies recommended. Patients also seek TCIM care for a variety of reasons, such as improving cancer outcomes, side effect and symptom management, holistic care, general wellbeing, and management of other health conditions,17 which results in more individualized and variable treatment.
In general, most treatments were recommended uniformly between the four cancer types. There are several reasons this consistency between cancer types may exist. Firstly, many patients seek TCIM care for symptom management and general wellbeing, where treatments may be less dependent on cancer type. These treatments may target an underlying physiological process independent of cancer type, such as malnutrition, cachexia/sarcopenia, nausea, or immune function. Secondly, some treatments have demonstrated benefit across a variety of cancer types. For example, exercise improves QOL and symptom management across various advanced cancers,18 and many NHPs, such as vitamin D,19 Trametes versicolor,20 and melatonin,21 have demonstrated benefit for cancer outcomes among various cancers. Finally, some natural products have a large body of preclinical evidence demonstrating activity against diverse targets of cancer growth and metastasis which are common among various cancer types, such as curcumin22,23 and green tea extract.24,25 In this scenario, if safety has been established in humans and limited clinical data are available, it is possible such treatments may be recommended across cancer types.
Although there were no strong associations between any treatments and cancer type, more than 50% of the NHPs analyzed showed statistically significant non-uniformity between cancer types. This suggests there may be some specificity in the care provided by NDs in this domain. For some of the differences found in NHP recommendations, there are reasonable justifications. Participants with pancreatic cancer had a smaller overall number of NHP recommendations compared with other cancer types and the most significant negative residuals. This may be due to the higher incidence of digestive issues, cachexia, and sarcopenia,26 which could result in NDs recommending fewer oral supplements to avoid worsening digestion or decreasing food intake. Linum usitatissimum (flaxseed) recommendations were higher in breast cancer (and to a lesser degree ovarian cancer), likely due to the lignan content, which impacts estrogen metabolism and signaling.27–31 Digestive enzymes were more frequently recommended in pancreatic cancer, which is almost certainly due to the high incidence of exocrine pancreatic insufficiency and digestive upset in this population.32 Calcium and vitamin K were more common in those with breast cancer, which could be due to a higher rate of osteoporosis and fractures in this cohort.33 However, for other discrepancies, the reasons are not clear, and in most cases the differences may not be clinically meaningful.
Natural health products were the most common treatment modality used by NDs. While we cannot know the reasons why a specific NHP or category of NHPs was recommended, the frequency of use of certain products is likely dependent on a variety of factors, including patient concerns and preferences, available evidence, potential for pleiotropic effects, clinician experience, and feasibility. For example, many of the top recommendations, including vitamin D, curcumin, melatonin, omega 3 fatty acids, and Trametes versicolor, have evidence for improving a variety of outcomes in both cancer and non-cancer populations, which could explain their high frequency of use. Vitamin D has been shown to reduce cancer mortality,19 decrease opioid use and fatigue in a palliative cancer setting,34 and reduce depressive symptoms.35,36 Curcumin has been shown to improve QOL,36 reduce inflammation,36 and help treat a wide variety of cancer treatment-related side effects.37,38 Melatonin has some evidence for improved survival,21 but ample evidence to improve sleep.39 Omega 3 fatty acids can help maintain lean body mass and overall nutrition status40,41 and may prevent oxaliplatin-induced peripheral neuropathy.42 Trametes versicolor has evidence for improved survival and immunomodulatory effects.20,43–45 These NHPs have the potential to benefit many patients; thus, it is not surprising they were the most recommended. It is notable that the evidence for NHP use in cancer care is more robust for symptom and side effect management than cancer survival outcomes. Therefore, it is reasonable to think that NHPs are used more often for these outcomes. This is strengthened by the fact that the number of ND visits significantly correlated with the number of NHP recommendations, which aligns with NDs addressing new side effects or adapting their treatment plans to complement different conventional cancer treatments.
Dietary counselling was the second most common treatment modality and the most uniform across all cancer types. The most notable finding is the overwhelming number of dietary encouragements compared with discouragements. Emphasizing the benefits of certain foods instead of the harms of others (e.g., encouraging whole foods rather than discouraging processed foods) promotes a “gain-framed” approach, which is more effective when trying to elicit a behaviour change.46 Research has also shown that people who set approach-oriented goals are more likely to succeed than people who set avoidance-related goals.47 Additionally, people with advanced cancer are at a greater risk for malnutrition, cachexia, and sarcopenia;48,49 thus, an emphasis on restricting foods may be detrimental.
Many of the top dietary encouragements, namely protein foods, vegetables, eating behaviour changes and nutrition education, and fruits (Table 5), could represent a combination of the most important and/or the most deficient aspects of nutrition for people with cancer. Research has shown that, for those with cancer, protein intakes of less than 1.2 g/kg are associated with muscle wasting and intakes above 1.4 g/kg are associated with muscle maintenance.50 This is significantly higher than the recommended daily allowance for adults (approximately 0.8 g/kg)51 and thus supports the recommendation to increase protein intake. Data also indicate that neither Americans nor Canadians consume the recommended daily intakes for fruits and vegetables;52,53 thus, recommendations to increase consumption are in line with this finding. Additionally, Canadians and Americans are generally deficient in fibre,54,55 which could also contribute to the high incidence of vegetable and fruit recommendations. Naturopathic doctors also discussed eating behaviours and nutrition education with many participants. Given that NDs focus on encouragements, and adding foods to the diet has been shown to be more difficult than removing them,56 emphasizing eating behaviours and providing education can help develop a better relationship with food, which could help participants adopt dietary changes.57 Beyond these top recommendations, others were used more sparingly. This could be due to patient overwhelm. Research shows that focusing on a few dietary changes at a time improves the likelihood of patient compliance.56 Furthermore, given the unique nutritional needs and preferences of each person, it is reasonable to expect that nutrition guidance may be more varied between patients.
The top dietary discouragements, including added sugars, refined grains, red meat, and alcohol, are in line with the World Cancer Research Fund’s guidelines for cancer prevention.58 Raw foods were also heavily discouraged, likely to ease digestion for patients undergoing cancer treatments. In addition to the absolute number of discouraged foods being lower than those encouraged, these specific discouragements are not high in frequency compared with those most encouraged. While positive framing partially explains this, practitioners are also likely to only record dietary recommendations if a change is perceived as needed, especially for discouragements like reduced alcohol intake.
Recommendations for mental health, TCM, and hyperthermia were among the least common (Table 2). The low incidence of recommendations for TCM and hyperthermia is not surprising. Hyperthermia is only offered at a limited number of participating clinics, which decreases the likelihood that patients have access to this therapy. Although many studies support the use of TCM practices in patients with cancer, such as acupuncture, the strongest evidence is for the treatment of certain cancer-related symptoms9 that may not be experienced by all patients. Furthermore, not all NDs practice acupuncture or other TCM practices. The low incidence of mental health recommendations is unexpected given the holistic nature of naturopathic medicine, the wide variety of items included in this category, and the importance of mental health, especially in people with cancer. Research has shown that people with cancer are at an increased risk for mental health disorders and reduced general mental wellbeing.59–62 Mental health is a complex subject and defining what is or is not a recommendation to support mental health is difficult. For example, certain aspects of nutrition and exercise could also serve as mental health support but were not categorized as mental health recommendations in this study. Furthermore, it is well documented that there are often errors in chart notes, including discrepancies between what is documented versus what is discussed during an appointment.63–65 One qualitative study showed that, during 10 physician visits, social and emotional topics were discussed in each visit yet only documented 30% of the time.66 Discussion amongst the clinician-investigators related to this topic revealed that, at least among some practitioners, this aspect of care is often talked about with patients but not always charted. The lack of mental health recommendations we observed could indicate a practice gap in ND care, either due to a lack of discussion surrounding mental health or due to a lack of effective documentation.
A recent survey of NDs who provide supportive cancer care collected data on the self-reported frequency of use of different treatment modalities.14 A section of this survey asked NDs how often they “frequently” recommend or refer out for certain therapies. This presents a unique opportunity to compare NDs’ perception of their care and the documented frequencies of recommendations from chart notes. Although a rigorous comparison is not possible, given what NDs perceive as “frequent” is subjective and the survey was not restricted to late-stage cancers, some general correlations can be made. Of the 99 respondents in the survey, 95% reported they frequently discussed nutrition and nutritional supplements, 63% reported they frequently recommended exercise, 46% reported they frequently recommended acupuncture or TCM, and 39% reported they frequently recommended OTC medications. This aligns with the data we collected on how often these items are recommended in practice. Two items contradict our data. First, 45% of NDs reported they frequently recommended IV and infusion therapy; however, over 80% of our cohort were recommended these therapies. This is almost certainly due to differences in patient populations; IV therapies are likely recommended far less frequently to patients with early stage or curative cancers, which would decrease the overall frequency of these recommendations in a survey that did not distinguish between cancer stages. Secondly, 72% of NDs reported they frequently recommended mind–body therapies, and 63% reported they frequently recommended or referred out for mental health counselling, whereas only 33% of our cohort were given recommendations pertaining to their mental health. This strengthens our hypothesis that there may be a gap in what is discussed versus what is documented, or it suggests that NDs do not discuss mental health as often as they believe.
Strengths and Limitations
To our knowledge, this is the largest study which comprehensively describes naturopathic treatment recommendations to patients with advanced cancers. The amount of data collected is novel in this field and the documentation we present describes the diverse and comprehensive nature of naturopathic oncology care across North America.
One prominent limitation was our ability to effectively categorize and summarize the data we collected. Firstly, clinical judgment was required from study clinician-investigators for most recommendations. For example, many NHPs are commercial or combination products, and clinical judgment was used to categorize these as separate compounds or a single product. Secondly, the intent of the treating ND when making a recommendation was not known, which can make categorizing treatments difficult. For example, if a recommendation was made to eat more fish, it was not clear whether the goal was to increase omega 3 intake, increase protein, or decrease intake of other forms of animal-protein. Finally, some items, such as yoga therapy, conceivably fit into multiple categories (i.e., mental health and exercise).
Other limitations include variations in licensure, scope of practice, and access to equipment between provinces and states, which inherently reduces the overall incidence of many recommendations such as IV therapies, prescription drugs, and hyperthermia. Additionally, data regarding adherence for these therapies were not collected, and thus the degree to which participants followed the guidance provided by their ND is unknown. Finally, since we did not capture the intention behind the use of each recommendation, our ability to interpret the findings is limited.
CONCLUSION
This observational study provides a comprehensive overview of naturopathic treatment recommendations for patients with late-stage breast, colorectal, ovarian, and pancreatic cancers in North America. Naturopathic doctors recommend a diverse array of treatments, highlighting the individualized nature of naturopathic care. Despite the large variety of treatments overall, the frequency with which several interventions are used is notable, suggesting some degree of consistency of care. Furthermore, there were no treatments recommended for one cancer type appreciably more than all others; however, minor differences were demonstrated, particularly with NHPs. Frequently, patients are recommended several NHPs, including vitamins, minerals, and herbal medicines. Intravenous vitamin C is commonly recommended, most patients are encouraged to exercise, and naturopathic doctors routinely give nutrition counselling using a positive framing approach that emphasizes the encouragement of certain foods and eating behaviours over discouragement. Mental health recommendations were low across all cancer types, which could indicate a practice gap or documentation issue. The data presented offer insights into the types of treatments used by naturopathic doctors and their consistency of use across different cancer types. These findings will enhance patients’ and healthcare providers’ understanding of the care provided by naturopathic doctors and inform future research avenues.
AUTHOR AFFILIATIONS
1Patterson Institute for Integrative Oncology Research, ON, Canada;
2The Centre for Health Innovation, ON, Canada;
3Bastyr University Research Institute, WA, USA;
4Integrated Health Clinic Cancer Care Centre, BC, Canada;
5Marsden Centre for Excellence in Integrative Medicine, ON, Canada;
6Health Source Integrative Medical Centre, ON, Canada;
7Naturopathic Specialists, LLC, AZ, USA;
8Salish Cancer Centre, WA, USA;
9Hawaii Integrative Oncology – Lokahi Health Centre, HI, USA;
10Tree of Health Integrative Medicine, WA, USA;
11Ottawa Hospital Research Institute, ON, Canada
ACKNOWLEDGEMENTS
Not applicable
CONFLICTS OF INTEREST DISCLOSURE
We have read and understood the CAND Journal’s policy on conflicts of interest and declare that we have none.
FUNDING
This study was funded by the Lotte and John Hecht Memorial Foundation.
REFERENCES
1. Horneber M, Bueschel G, Dennert G, Less D, Ritter E, Zwahlen M. How many cancer patients use complementary and alternative medicine: a systematic review and meta-analysis. Integr Cancer Ther. 2012;11(3):187–203. https://doi.org/10.1177/1534735411423920
Crossref
2. Wortmann JK, Bremer A, Eich HT, et al. Use of complementary and alternative medicine by patients with cancer: a cross-sectional study at different points of cancer care. Med Oncol. 2016;33(7):78–78. https://doi.org/10.1007/s12032-016-0790-4
Crossref PubMed
3. Judson PL, Abdallah R, Xiong Y, Ebbert J, Lancaster JM. Complementary and alternative medicine use in individuals presenting for care at a comprehensive cancer center. Integr Cancer Ther. 2016;16(1):96–103. https://doi.org/10.1177/1534735416660384
Crossref PubMed PMC
4. Steel A, Foley H, Bradley R, et al. Overview of international naturopathic practice and patient characteristics: results from a cross-sectional study in 14 countries. BMC Complement Med Ther. 2020;20(1):59. https://doi.org/10.1186/s12906-020-2851-7
Crossref PubMed PMC
5. Snider P, Zeff J. Unifying principles of naturopathic medicine origins and definitions. Integr Med (Encinitas). 2019;18(4):36–39.
6. Steel A, Redmond R, Schloss J, et al. International prevalence of consultation with a naturopathic practitioner: a systematic review and meta-analysis. BMJ Open. 2022;12(7):e056075. https://doi.org/10.1136/bmjopen-2021-056075
Crossref PubMed PMC
7. Marsden E, Nigh G, Birdsall S, Wright H, Traub M. Oncology Association of Naturopathic Physicians: principles of care guidelines. Curr Oncol. 2019;26(1):12–18. https://doi.org/10.3747/co.26.4815
Crossref PubMed PMC
8. Oncology Association of Naturopathic Physicians. FABNO certification. Accessed April 7, 2023, at https://oncanp.org/fabno-certification/
9. Greenlee H, DuPont-Reyes MJ, Balneaves LG, et al. Clinical practice guidelines on the evidence-based use of integrative therapies during and after breast cancer treatment. CA Cancer J Clin. 2017;67(3):194–232. https://doi.org/10.3322/caac.21397
Crossref PubMed PMC
10. Carlson LE, Ismaila N, Addington EL, et al. Integrative oncology care of symptoms of anxiety and depression in adults with cancer: Society for Integrative Oncology-ASCO guideline. J Clin Oncol. 2023;41(28):4562–4591. https://doi.org/10.1200/jco.23.00857
Crossref PubMed
11. Standish LJ, Dowd F, Sweet E, et al. Breast cancer integrative oncology care and its costs. Integr Cancer Ther. 2017;16(1):85–95. https://doi.org/10.1177/1534735416649034
Crossref
12. Standish LJ, Greene K, Greenlee H, Kim JG, Grosshans C. Complementary and alternative medical treatment of breast cancer: a survey of licensed North American naturopathic physicians. Altern Ther Health Med. 2002;8(5):68–70; 72–75.
PubMed
13. Seely D, Ennis JK, McDonell E, Zhao L. Naturopathic oncology care for thoracic cancers: a practice survey. Integr Cancer Ther. 2018;17(3):793–805. https://doi.org/10.1177/1534735418759420
Crossref PubMed PMC
14. Rizzolo E, Legacy M, Conte E, Elsayed M, Seely D. Advancing naturopathic cancer care: a survey of naturopathic doctors to identify practice patterns and knowledge gaps (Manuscript submitted for publication). CAND J. 2025;32(1):196. https://doi.org/10.54434/candj.196
Crossref
15. Standish LJ, Sweet E, Chiang PF. Naturopathic oncology for advanced cancers: Survival outcomes from the Canadian/US integrative oncology study. CAND Journal. 2025;32(2):4–20. https://doi.org/10.54434/candj.206
16. Conte E, Rizzolo E, Flower G, Legacy M, Seely D. Intravenous vitamin C in cancer care: evidence review and practical guidance for integrative oncology practitioners. CAND J. 2024;31(1):2–18. https://doi.org/10.54434/candj.149
Crossref
17. Keene MR, Heslop IM, Sabesan SS, Glass BD. Complementary and alternative medicine use in cancer: a systematic review. Complement Ther Clin Pract. 2019;35:33–47. https://doi.org/10.1016/j.ctcp.2019.01.004
Crossref PubMed
18. Rodríguez-Cañamero S, Cobo-Cuenca AI, Carmona-Torres JM, et al. Impact of physical exercise in advanced-stage cancer patients: systematic review and meta-analysis. Cancer Med. 2022;11(19):3714–3727. https://doi.org/10.1002/cam4.4746
Crossref PubMed PMC
19. Guo Z, Huang M, Fan D, et al. Association between vitamin D supplementation and cancer incidence and mortality: a trial sequential meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. 2023;63(26):8428–8442. https://doi.org/10.1080/10408398.2022.2056574
Crossref
20. Zhong L, Yan P, Lam WC, Yao L, Bian Z. Coriolus versicolor and Ganoderma lucidum related natural products as an adjunct therapy for cancers: a systematic review and meta-analysis of randomized controlled trials. Front Pharmacol. 2019;10:703. https://doi.org/10.3389/fphar.2019.00703
Crossref PubMed PMC
21. Seely D, Wu P, Fritz H, et al. Melatonin as adjuvant cancer care with and without chemotherapy: a systematic review and meta-analysis of randomized trials. Integr Cancer Ther. 2012;11(4):293–303. https://doi.org/10.1177/1534735411425484
Crossref
22. Wang W, Li M, Wang L, Chen L, Goh BC. Curcumin in cancer therapy: exploring molecular mechanisms and overcoming clinical challenges. Cancer Lett. 2023;570:216332. https://doi.org/10.1016/j.canlet.2023.216332
Crossref PubMed
23. Kumar G, Mittal S, Sak K, Tuli HS. Molecular mechanisms underlying chemopreventive potential of curcumin: current challenges and future perspectives. Life Sci. 2016;148:313–328. https://doi.org/10.1016/j.lfs.2016.02.022
Crossref PubMed
24. Min KJ, Kwon TK. Anticancer effects and molecular mechanisms of epigallocatechin-3-gallate. Integr Med Res. 2014;3(1):16–24. https://doi.org/10.1016/j.imr.2013.12.001
Crossref PubMed PMC
25. Almatroodi SA, Almatroudi A, Khan AA, Alhumaydhi FA, Alsahli MA, Rahmani AH. Potential therapeutic targets of epigallocatechin gallate (EGCG), the most abundant catechin in green tea, and its role in the therapy of various types of cancer. Molecules. 2020;25(14). https://doi.org/10.3390/molecules25143146
Crossref
26. Hou Y-C, Chen C-Y, Huang C-J, et al. The differential clinical impacts of cachexia and sarcopenia on the prognosis of advanced pancreatic cancer. Cancers. 2022;14(13):3137.
Crossref PubMed PMC
27. Thompson LU, Chen JM, Li T, Strasser-Weippl K, Goss PE. Dietary flaxseed alters tumor biological markers in postmenopausal breast cancer. Clin Cancer Res. 2005;11(10):3828–3835. https://doi.org/10.1158/1078-0432.Ccr-04-2326
Crossref PubMed
28. Brooks JD, Ward WE, Lewis JE, et al. Supplementation with flaxseed alters estrogen metabolism in postmenopausal women to a greater extent than does supplementation with an equal amount of soy. Am J Clin Nutr. 2004;79(2):318–325. https://doi.org/10.1093/ajcn/79.2.318
Crossref PubMed
29. Ansenberger K, Richards C, Zhuge Y, et al. Decreased severity of ovarian cancer and increased survival in hens fed a flaxseed-enriched diet for 1 year. Gynecol Oncol. 2010;117(2):341–7. https://doi.org/10.1016/j.ygyno.2010.01.021
Crossref PubMed PMC
30. Dikshit A, Hales K, Hales DB. Whole flaxseed diet alters estrogen metabolism to promote 2-methoxtestradiol-induced apoptosis in hen ovarian cancer. J Nutr Biochem. 2017;42:117–125. https://doi.org/10.1016/j.jnutbio.2017.01.002
Crossref PubMed PMC
31. Calado A, Neves PM, Santos T, Ravasco P. The effect of flaxseed in breast cancer: a literature review. Front Nutr. 2018;5:4. https://doi.org/10.3389/fnut.2018.00004
Crossref PubMed PMC
32. Roeyen G, Berrevoet F, Borbath I, et al. Expert opinion on management of pancreatic exocrine insufficiency in pancreatic cancer. ESMO Open. 2022;7(1):100386. https://doi.org/10.1016/j.esmoop.2022.100386
Crossref PubMed PMC
33. Lee S, Yoo JI, Lee YK, et al. Risk of osteoporotic fracture in patients with breast cancer: meta-analysis. J Bone Metab. 2020;27(1):27–34. https://doi.org/10.11005/jbm.2020.27.1.27
Crossref PubMed PMC
34. Helde Frankling M, Klasson C, Sandberg C, et al. ‘Palliative-D’-vitamin D supplementation to palliative cancer patients: a double blind, randomized placebo-controlled multicenter trial. Cancers (Basel). 2021;13(15):3707. https://doi.org/10.3390/cancers13153707
Crossref PubMed PMC
35. Ghaemi S, Zeraattalab-Motlagh S, Jayedi A, Shab-Bidar S. The effect of vitamin D supplementation on depression: a systematic review and dose-response meta-analysis of randomized controlled trials. Psychol Med. 2024;54(15):1–10. https://doi.org/10.1017/s0033291724001697
Crossref PubMed PMC
36. Wang R, Xu F, Xia X, et al. The effect of vitamin D supplementation on primary depression: a meta-analysis. J Affect Disord. 2024;344:653–661. https://doi.org/10.1016/j.jad.2023.10.021
Crossref
37. Panahi Y, Saberi-Karimian M, Valizadeh O, et al. Effects of curcuminoids on systemic inflammation and quality of life in patients with colorectal cancer undergoing chemotherapy: a randomized controlled trial. Adv Exp Med Biol. 2021;1328:1–9. https://doi.org/10.1007/978-3-030-73234-9_1
Crossref
38. Dharman S, G M, Shanmugasundaram K, Sampath RK. A systematic review and meta-analysis on the efficacy of curcumin/turmeric for the prevention and amelioration of radiotherapy/radiochemotherapy induced oral mucositis in head and neck cancer patients. Asian Pac J Cancer Prev. 2021;22(6):1671–1684. https://doi.org/10.31557/apjcp.2021.22.6.1671
Crossref PubMed PMC
39. Cruz-Sanabria F, Bruno S, Crippa A, et al. Optimizing the time and dose of melatonin as a sleep-promoting drug: a systematic review of randomized controlled trials and dose-response meta-analysis. J Pineal Res. 2024;76(5):e12985. https://doi.org/10.1111/jpi.12985
Crossref PubMed
40. Shirai Y, Okugawa Y, Hishida A, et al. Fish oil-enriched nutrition combined with systemic chemotherapy for gastrointestinal cancer patients with cancer cachexia. Sci Rep. 2017;7(1):4826. https://doi.org/10.1038/s41598-017-05278-0
Crossref PubMed PMC
41. Tao X, Zhou Q, Rao Z. Efficacy of ω-3 polyunsaturated fatty acids in patients with lung cancer undergoing radiotherapy and chemotherapy: a meta-analysis. Int J Clin Pract. 2022;2022:6564466. https://doi.org/10.1155/2022/6564466
Crossref
42. Zhang X, Chen H, Lu Y, et al. Prevention of oxaliplatin-related neurotoxicity by ω-3 PUFAs: A double-blind randomized study of patients receiving oxaliplatin combined with capecitabine for colon cancer. Medicine. 2020;99(50):e23564. https://doi.org/10.1097/md.0000000000023564
Crossref
43. Pilkington K, Wieland LS, Teng L, Jin XY, Storey D, Liu JP. Coriolus (Trametes) versicolor mushroom to reduce adverse effects from chemotherapy or radiotherapy in people with colorectal cancer. Cochrane Database Syst Rev. 2022;11(11):CD012053. https://doi.org/10.1002/14651858.CD012053.pub2
PubMed PMC
44. Saleh MH, Rashedi I, Keating A. Immunomodulatory properties of Coriolus versicolor: the role of polysaccharopeptide. Front Immunol. 2017;8:1087.
Crossref PubMed PMC
45. Eliza WL, Fai CK, Chung LP. Efficacy of Yun Zhi (Coriolus versicolor) on survival in cancer patients: systematic review and meta-analysis. Recent Pat Inflamm Allergy Drug Discov. 2012;6(1):78–87. https://doi.org/10.2174/187221312798889310
Crossref
46. Mikels JA, Young NA, Liu X, Stine-Morrow EAL. Getting to the heart of the matter in later life: the central role of affect in health message framing. Gerontologist. 2021;61(5):756–762. https://doi.org/10.1093/geront/gnaa128
Crossref PMC
47. Oscarsson M, Carlbring P, Andersson G, Rozental A. A large-scale experiment on New Year’s resolutions: approach-oriented goals are more successful than avoidance-oriented goals. PLOS One. 2020;15(12):e0234097. https://doi.org/10.1371/journal.pone.0234097
Crossref PMC
48. Surov A, Wienke A. Prevalence of sarcopenia in patients with solid tumors: a meta-analysis based on 81,814 patients. JPEN J Parenter Enteral Nutr. 2022;46(8):1761–1768. https://doi.org/10.1002/jpen.2415
Crossref PubMed
49. Mariean CR, Tiucă OM, Mariean A, Cotoi OS. Cancer cachexia: new insights and future directions. Cancers (Basel). 2023;15(23). https://doi.org/10.3390/cancers15235590
Crossref PubMed PMC
50. Capitão C, Coutinho D, Neves PM, et al. Protein intake and muscle mass maintenance in patients with cancer types with high prevalence of sarcopenia: a systematic review. Support Care Cancer. 2022;30(4):3007–3015. https://doi.org/10.1007/s00520-021-06633-8
Crossref
51. Government of Canada. Dietary reference intakes tables: reference values for macronutrients [Table]. Accessed March 13, 2025, at https://www.canada.ca/en/health-canada/services/food-nutrition/healthy-eating/dietary-reference-intakes/tables/reference-values-macronutrients.html#a2
52. Lee SH, Moore LV, Park S, Harris DM, Blanck HM. Adults meeting fruit and vegetable intake recommendations – United States, 2019. MMWR Morb Mortal Weekly Rep. 2022;71(1):1–9. https://doi.org/10.15585/mmwr.mm7101a1
Crossref
53. Polsky JY, Garriguet D. Change in vegetable and fruit consumption in Canada between 2004 and 2015. Health Rep. 2020;31(4):3–12. https://doi.org/10.25318/82-003-x202000400001-eng
PubMed
54. Quagliani D, Felt-Gunderson P. Closing America’s fiber intake gap: communication strategies from a food and fiber summit. Am J Lifestyle Med. 2017;11(1):80–85. https://doi.org/10.1177/1559827615588079
Crossref
55. Government of Canada. Fibre. Accessed March 13, 2025, at https://www.canada.ca/en/health-canada/services/nutrients/fibre.html
56. Wang F, Cai H, Gu K, et al. Adherence to Dietary Recommendations among long-term breast cancer survivors and cancer outcome associations. Cancer Epidemiol Biomarkers Prev. 2020;29(2):386–395. https://doi.org/10.1158/1055-9965.Epi-19-0872
Crossref PMC
57. van’t Riet J, Sijtsema SJ, Dagevos H, De Bruijn GJ. The importance of habits in eating behaviour. An overview and recommendations for future research. Appetite. 2011;57(3):585–596. https://doi.org/10.1016/j.appet.2011.07.010
Crossref
58. World Cancer Research Fund. Cancer Prevention Recommendations. http://www.wcrf.org/int/research-we-fund/our-cancer-prevention-recommendations
59. Mosher CE, Winger JG, Given BA, Helft PR, O’Neil BH. Mental health outcomes during colorectal cancer survivorship: a review of the literature. Psychooncology. 2016;25(11):1261–1270. https://doi.org/10.1002/pon.3954
Crossref PubMed PMC
60. Carreira H, Williams R, Müller M, Harewood R, Stanway S, Bhaskaran K. Associations between breast cancer survivorship and adverse mental health outcomes: a systematic review. J Natl Cancer Inst. 2018;110(12):1311–1327. https://doi.org/10.1093/jnci/djy177
Crossref PubMed PMC
61. Hu S, Baraghoshi D, Chang CP, et al. Mental health disorders among ovarian cancer survivors in a population-based cohort. Cancer Med. 2023;12(2):1801–1812. https://doi.org/10.1002/cam4.4976
Crossref PMC
62. Yi JC, Syrjala KL. Anxiety and depression in cancer survivors. Med Clin North Am. 2017;101(6):1099–1113. https://doi.org/10.1016/j.mcna.2017.06.005
Crossref PubMed PMC
63. Bell SK, Delbanco T, Elmore JG, et al. Frequency and types of patient-reported errors in electronic health record ambulatory care notes. JAMA Netw Open. 2020;3(6):e205867. https://doi.org/10.1001/jamanetworkopen.2020.5867
Crossref PubMed PMC
64. Song J, Zolnoori M, Scharp D, et al. Do nurses document all discussions of patient problems and nursing interventions in the electronic health record? A pilot study in home healthcare. JAMIA Open. 2022;5(2):ooac034. https://doi.org/10.1093/jamiaopen/ooac034
Crossref PubMed PMC
65. Turer CB, Barlow SE, Montaño S, Flores G. Discrepancies in communication versus documentation of weight-management benchmarks: analysis of recorded visits with Latino children and associated health-record documentation. Glob Pediatr Health. 2017;4:2333794x16685190. https://doi.org/10.1177/2333794x16685190
Crossref PubMed PMC
66. Prater L, Sanchez A, Modan G, et al. Electronic health record documentation patterns of recorded primary care visits focused on complex communication: a qualitative study. Appl Clin Inform. 2019;10(2):247–253. https://doi.org/10.1055/s-0039-1683986
Crossref PubMed PMC
APPENDIX A: STATISTICAL ANALYSES
TABLE A1 Chi-Squared Analyses for Natural Health Product Recommendations
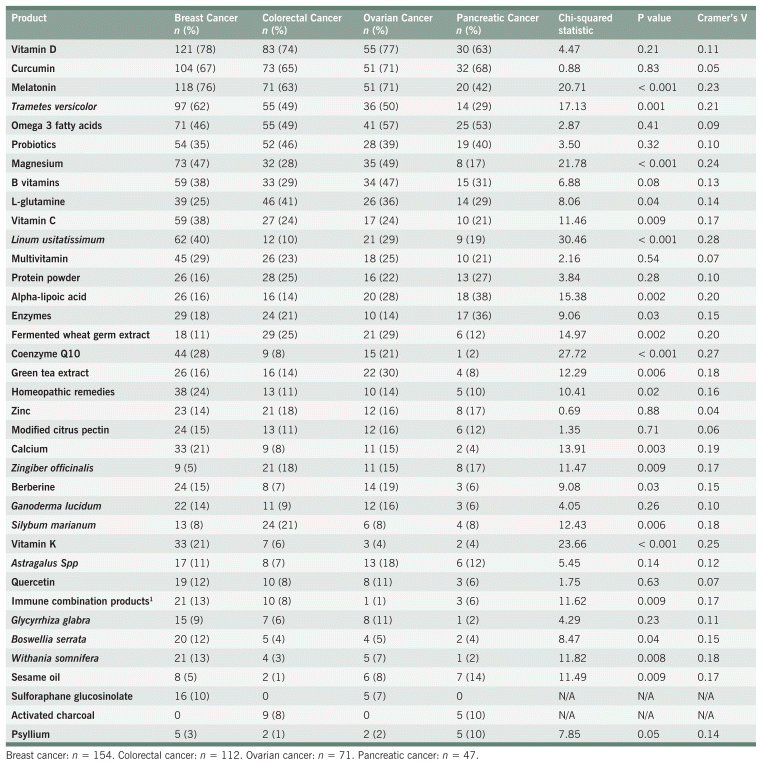
TABLE A2 Standardized Residuals for Natural Health Product Recommendations
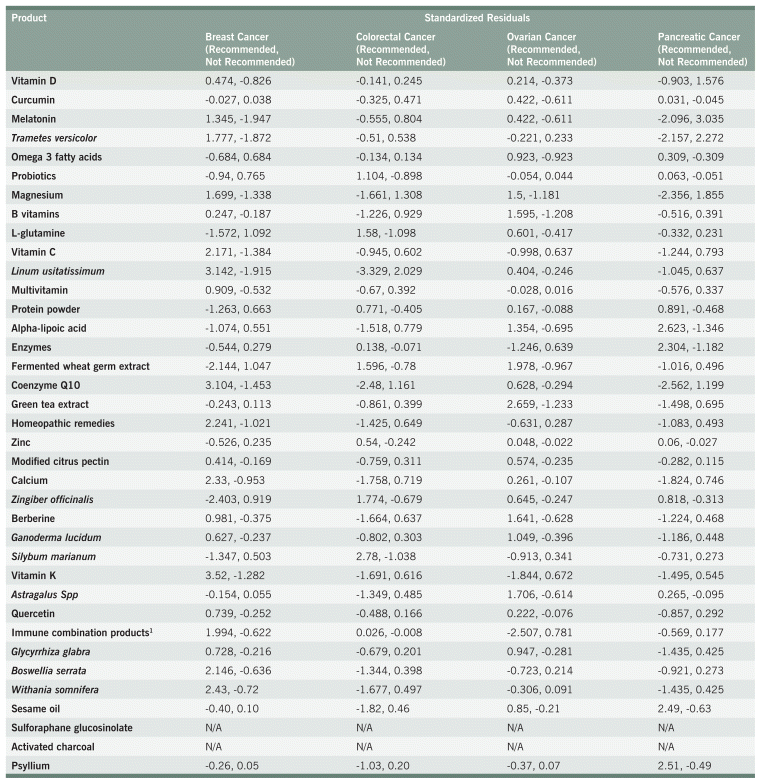
TABLE A3 Chi-Squared Analyses for Nutrition Encouragement Recommendations
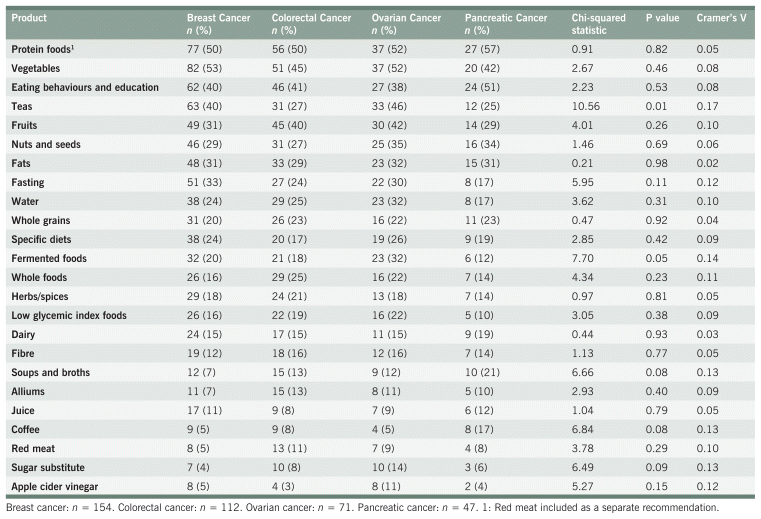
TABLE A4 Standardized Residuals for Nutrition Encouragement Recommendations
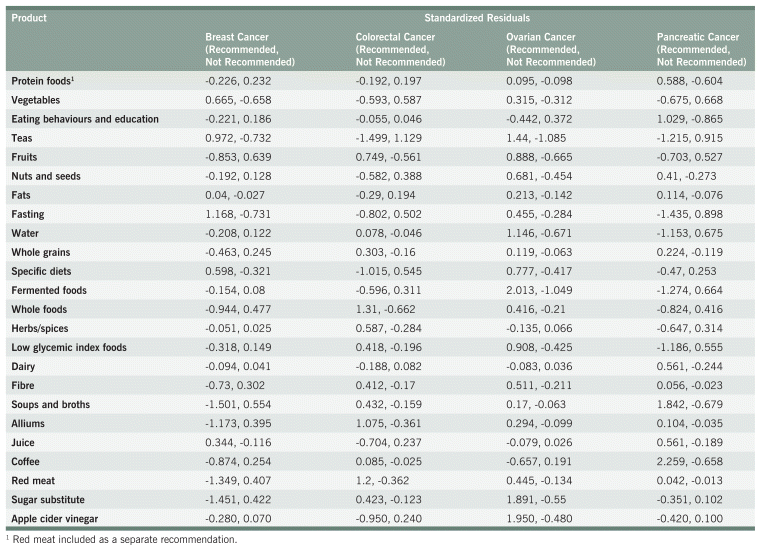
TABLE A5 Chi-Squared Analyses for Nutrition Discouragement Recommendations
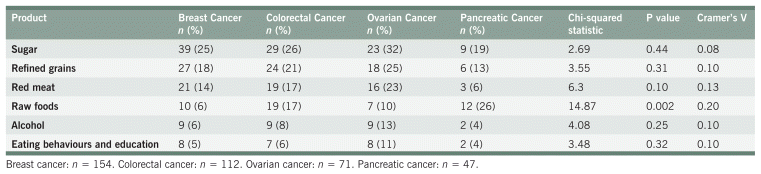
TABLE A6 Standardized Residuals for Nutrition Discouragement Recommendations
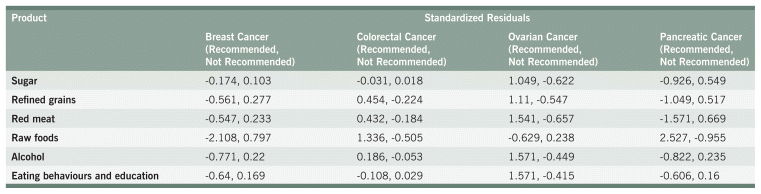
TABLE A7 Chi-Squared Analyses for Parenteral Recommendations
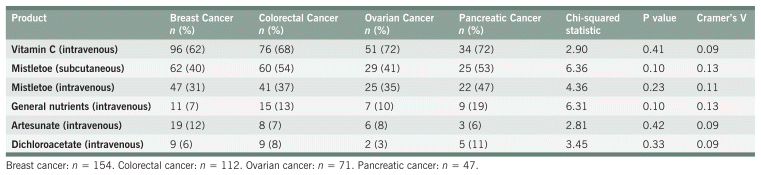
TABLE A8 Standardized Residuals for Parenteral Recommendations
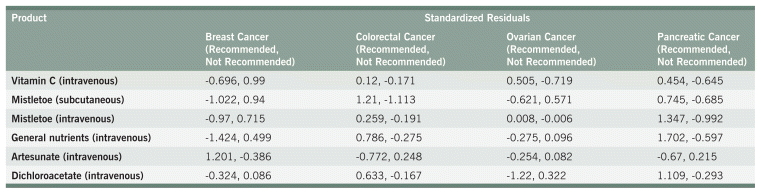
TABLE A9 Chi-Squared Analyses for Other Common Recommendations
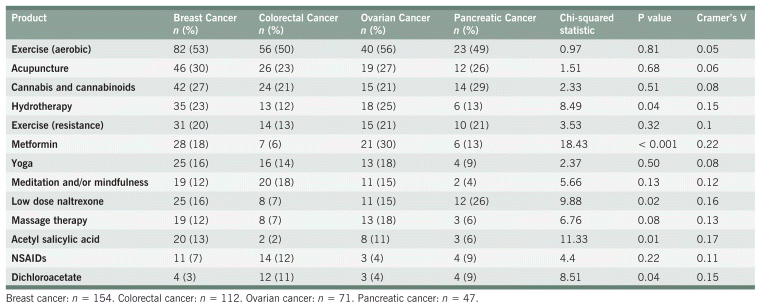
TABLE A10 Standardized Residuals for Other Common Recommendations
Correspondence to: Dugald Seely, ND, MSc, 429 MacLaren Street, Ottawa, ON K2P 0M7, Canada. E-mail: dseely@thechi.ca
To cite: Seely D, Legacy M, Conte E, et al. A comprehensive description of naturopathic care for advanced cancers: outcomes from the Canadian/US integrative oncology study. CAND Journal. 2025;32(2):21-40. https://doi.org/10.54434/candj.207
Received: 26 March 2025; Accepted: 2 May 2025; Published: 19 June 2025
© Author(s) 2025. This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution-NonCommercial (CC BY-NC 4.0) license. See https://creativecommons.org/licenses/by-nc/4.0/. For commercial re-use, please contact candj@cand.ca.
Published by the Canadian Association of Naturopathic Doctors.
CAND Journal | Volume 32, No. 2, June 2025
(Return to Top)