Original Article
Naturopathic Oncology for Advanced Cancers: Survival Outcomes from the Canadian/US Integrative Oncology Study
Leanna J. Standish,1 ND, PhD, LAc, Erin Sweet,1 ND, MPH, Peih F. Chiang,1 ND, Mark Legacy,2,3 CCRP, Ellen Conte,2 ND, Erica Rizzolo,2,3 ND, Linda Dale,1 CCRP, Athanasios Psihogios,2 MPH, Julie Ennis,2 PhD, Gurdev Parmar,4 ND, Eric Marsden,5 ND, Michael Reid,6 ND, Dan Rubin,7 ND, Paul Reilly,8 ND, LAc, Michael Traub,9 ND, Eleonora Naydis,10 ND, LAc, Tim Ramsay,11 PhD, and Dugald Seely,2,3 ND, MSc
ABSTRACT
Background: Several traditional, complementary, and integrative therapies have been studied for their effect on cancer outcomes; however, few studies have evaluated naturopathic oncology in a real-world setting. We conducted an observational cohort study to evaluate whether consulting with a naturopathic doctor (ND) improves survival in people with advanced cancers.
Methods: Participants with metastatic breast, metastatic colorectal, advanced ovarian, or advanced pancreatic cancer were recruited from 12 North American naturopathic clinics. The primary outcome was 5-year overall survival compared with a matched cohort from the Surveillance Epidemiology and End Results (SEER) registry. We additionally evaluated survival based on the number of ND visits and intravenous vitamin C (IVC), intravenous mistletoe, and hyperthermia treatments.
Results: 400 participants were enrolled: 165 had breast cancer, 116 had colorectal cancer, 72 had ovarian cancer, and 47 had pancreatic cancer. A bootstrapped analysis showed no significant differences in 5-year survival compared with SEER (breast: median hazard ratio [HR] 1.10, 95% confidence interval [CI] 0.80–1.54; colorectal: median HR 0.95, 95% CI 0.68–1.31; ovarian: median HR 1.08, 95% CI 0.64–1.86; pancreatic: median HR 0.76, 95% CI 0.51–1.13). Higher survival odds were seen with increasing IVC treatments in breast cancer, and with increasing ND visits in all cancer types except ovarian.
Conclusion: No survival benefits were seen in patients with advanced cancers who saw an ND compared with registry data. After controlling for survivorship bias, in general, the number of ND visits positively correlated with improved survival; however, the number of select naturopathic treatments did not. Findings should be interpreted with caution given study limitations.
Key Words Naturopathic medicine, integrative medicine, integrative oncology, naturopathic care, cancer survival, naturopathic doctor, naturopathic physician
INTRODUCTION
Despite advances in cancer screening and treatment, mortality remains high. In 2022, nearly 10 million people worldwide died from cancer.1 In Canada and the United States, cancer is the first and second leading cause of death, respectively.2,3 Innovative approaches are needed to improve survival rates.
Traditional, complementary, and integrative medicine (TCIM) is commonly used by people with cancer, with rates of use ranging from 40 to 80%.4–6 In North America, naturopathic medicine is a system of care provided by naturopathic doctors (NDs) that is recognized as part of the TCIM umbrella and includes the use of nutritional advice, dietary supplements, botanical medicine, nutrient and phytopharmaceutical injection therapy, acupuncture, counselling and emotional support, and lifestyle modifications.7 Naturopathic medicine is one of the most commonly practiced systems of TCIM in the Western world,8 with approximately 8,000 licensed NDs in North America.9 Studies suggest naturopathic medicine use is higher among those with cancer than the general population.8
Existing research on naturopathic medicine in cancer has typically focused on specific therapies, and outcomes related to quality of life (QOL) and symptom burden over survival.10 Some interventions have demonstrated improvements in survival for patients with cancer, including Trametes versicolor (turkey tail mushroom),11 Viscum album (mistletoe extract),12 exercise,13 and the Mediterranean Diet.14,15 Although evidence is increasing, most studies have focused on single agents in isolation, which does not accurately reflect naturopathic medicine’s holistic and multimodal real-world application in cancer care. Few studies have evaluated survival outcomes for patients with advanced cancer receiving naturopathic care in a pragmatic setting. Evidence supporting benefits is currently limited,16,17 highlighting the need for additional research.
The Canadian/US Integrative Oncology Study (CUSIOS) is an observational cohort study with the overarching goal of studying naturopathic oncology in a real-world setting. The primary objective was to measure survival in patients with late-stage breast, colorectal, ovarian, or pancreatic cancer who consulted with an ND. Five-year overall survival was compared with matched comparators from the Surveillance Epidemiology and End Results (SEER) database. Secondary objectives included describing the scope of treatments recommended to patients by NDs, comparing survival amongst enrolled participants based on the number of ND visits and the frequency of use of verifiably received naturopathic treatments, collecting health-related QOL data, estimating costs of naturopathic cancer care, and gathering information on participants’ qualitative experience of care. Data on the scope of ND recommendations, QOL, cost, and qualitative experiences are not discussed here and will be reported in separate publications.
In this paper, we compare 5-year survival outcomes of patients with advanced cancers who received naturopathic care with SEER as an external comparator. We also compare survival internally among enrolled participants based on the number of ND visits and naturopathic treatments received. We hypothesized that overall survival for participants enrolled in this study would be superior to what is reported by SEER.
METHODS
This paper was written alongside our reports on cost, QOL, qualitative experiences, and the type and frequency of ND recommendations to study participants.18 Many of the study methods were developed independently of the study outcomes; thus, there will be significant overlap.
Study Design
This was an observational study of patients with advanced breast, colorectal, ovarian, or pancreatic cancer who consulted an ND in North America. Recruitment and follow-up were primarily prospective; however, there were no restrictions based on date of diagnosis or enrollment date relative to the participant’s first visit with their ND. Thus, participants had varying levels of retrospective data collection. Clinics were requested to approach all eligible patients; however, we did not have a method to verify consecutive recruitment.
Setting
The study was conducted at 12 outpatient naturopathic clinics located in Canada (n = 5) and the United States (US) (n = 7). The primary coordinating centres were the Bastyr Center for Natural Health in Seattle, Washington, and The Centre for Health Innovation in Ottawa, Ontario. Additional sites were: Integrated Health Clinic Cancer Care Centre, Fort Langley, British Columbia; Marsden Centre for Excellence in Integrative Medicine, Vaughan, Ontario; Health Source Integrative Medical Centre, Kitchener, Ontario; Vital Victoria Naturopathic Clinic, Victoria, British Columbia; Naturopathic Specialists LLC, Scottsdale, Arizona; Salish Care Center, Fife, Washington; Hawaii Integrative Oncology, Kailua-Kona, Hawaii; Tree of Health Integrative Medicine, Woodinville, Washington; Seattle Integrative Cancer Center, Seattle, Washington; Advanced Integrative Medical Science Institute, Seattle, Washington. Clinics were selected for their focus in cancer care and NDs with a depth of experience in the field. A total of 29 NDs provided care to at least one study participant. All NDs had experience in a cancer care setting, and 15 were Fellows of the American Board of Naturopathic Oncology.
Participants
Eligible participants were adults 18 years of age or older who presented for care to a participating clinic with a confirmed diagnosis of one of the following:
- ▪ Metastatic breast cancer
(de novo stage IV or recurrent)
- ▪ Metastatic colorectal cancer
(de novo stage IV or recurrent)
- ▪ Advanced ovarian cancer
(stage III or IV, de novo or recurrent)
- ▪ Advanced pancreatic cancer
(stage III or IV, de novo or recurrent)
Stages were determined per the American Joint Committee on Cancer’s staging guidelines (version 7). Participants additionally had to be a citizen or permanent resident of the country in which they were receiving treatment. All participants signed an informed consent form prior to participating. Recruitment began in June 2015 and concluded in March 2020. Participants were followed for at least 2 years and up to 3 years. Variable follow-up time occurred due to funding restraints, which arose due to a longer than anticipated recruitment period. All participants were offered a $200 stipend upon enrolment to be used towards any practitioner consultation at the participant’s naturopathic clinic.
Naturopathic Care
Participants received treatment from their ND independent of this research study. All recommendations made by NDs to patients on this study were collected and are reported in a separate manuscript.18 In brief, recommendations spanned various modalities of treatment within the ND scope of practice, including natural health products, nutrition advice, intravenous (IV) and injectable therapies, physical medicine and body-based therapies, prescription and over-the-counter medications, mental health, Traditional Chinese Medicine, and hyperthermia.
Regulatory Adherence
Ethics approval was obtained from the Research Ethics Board of the Canadian College of Naturopathic Medicine and the Institutional Review Board at the Office of Research Integrity at Bastyr University prior to study initiation. The study was registered with clinicaltrials.gov, NCT02494037.
Outcomes
The primary study outcome was 5-year overall survival compared with a matched cohort from the SEER database. As a secondary outcome, we compared survival rates among study participants based on the number of ND visits and the frequency of use of verifiably received naturopathic treatments, including intravenous vitamin C (IVC), IV mistletoe, and hyperthermia.
Data Sources
Cancer treatments and clinical data were collected from naturopathic clinic records and conventional medical records. Naturopathic clinic records were reviewed from the first visit until the study end date, and conventional records were reviewed from diagnosis until the study end date. Date of death was collected from medical chart review and obituary searches. For US participants, the National Death Index was also queried. Last known survival date was collected from medical records if survival status was unknown. SEER data were obtained from SEER*Stat 8.4 and included patients diagnosed from 2010 to 2019.
Sample Size
The total expected study sample size was 400 patients, of which it was estimated 150 would have breast cancer, 150 would have colorectal cancer, 50 would have ovarian cancer, and 50 would have pancreatic cancer. The sample size was determined based on pragmatic considerations related to the number of estimated eligible patients the sites would see, the expected rate of enrollment, study duration, and available funding.
Quantitative Variables
Survival time for our population was defined as the date of stage III or IV diagnosis until study completion. Participants who were diagnosed at stage III but progressed to stage IV prior to enrolment were classified as stage IV; participants who were diagnosed at stage III but progressed to stage IV after enrolment were classified as stage III. Study completion was defined as occurring 3 years after a participant’s first visit to a study clinic, death, end of study, or date of last known survival. Participants were only included in the primary survival analysis if their date of diagnosis occurred in 2010 or later, as SEER does not report some key variables prior to 2010. In cases where the date of diagnosis was not fully known (i.e., only month or year), the dates were adjusted using the “Date of Diagnosis” algorithm described in the SEER Program Coding and Staging Manual 2023.19 Although 5-year overall survival was the primary outcome, some patients had survival times longer than 5 years depending on the lag time between date of diagnosis and the first visit with their ND. For these participants, survival time was truncated at 5 years.
Naturopathic treatments were abstracted from naturopathic clinic records and separated into distinct categories using the Research Electronic Data Capture (REDCap) platform. Entries were verified by a second team member. Variables for the internal survival comparison included the number of visits with an ND, and the number of IVC, IV mistletoe, and hyperthermia treatments. These treatments were chosen because they were more invasive and costly relative to other naturopathic oncology treatments, they had a high frequency of use in our population, and we were able to assess compliance given they were administered in-clinic. Data were only included if we could verifiably determine that participants received treatments at one of the study clinics (i.e., data on treatments received at other clinics or centres were not collected).
Statistical Methods
To analyze 5-year survival, we compared study participants with matched comparators from the SEER registry using sex, age, year of diagnosis, histology, hormone receptor status (breast cancer only), and cancer stage as matching variables. Each cancer type was analyzed separately. Participants were matched on all applicable variables and, additionally, were only matched if their lag time between date of diagnosis and date of study enrolment was less than a SEER patient’s survival time. Survival was analyzed using a bootstrap method. One randomly selected matched SEER patient was included for each study participant to create a cohort of equal size. A hazard ratio (HR), confidence interval (CI), and P value were generated using the Cox proportional hazards model. This analysis was run 10,000 times, each time randomly selecting one matched SEER patient for each study participant. The median HR and CI for all 10,000 outputs were used to determine statistical significance. We additionally conducted post-hoc sub-group analyses for each cancer type. In the first, we only included study participants whose lag time was less than or equal to the median lag time of participants with that cancer type (i.e., short lag time). In the second, we only included study participants who were diagnosed with de novo stage III or IV disease. Ovarian cancer was omitted from this analysis given more than 90% of participants were diagnosed de novo. In the third, we only included study participants who had a short lag time and were diagnosed de novo. Median lag times for each cancer type were as follows:
- ▪ Breast: 6 months
- ▪ Colorectal: 6 months
- ▪ Ovarian: 8 months
- ▪ Pancreatic: 2 months
Bootstrapped analyses are meant to generate a distribution of statistics to estimate confidence intervals. Calculating a single P value is therefore not necessary and provides less information than the confidence intervals. Results were considered significant if the confidence interval did not cross 1.00. Hazard ratios < 1.00 indicate a beneficial effect for study participants.
Independently of the bootstrapped analyses, Kaplan-Meier curves were created to visually represent the data for each cancer type. Each curve contained three cohorts: (1) all SEER patients with the appropriate cancer type (SEER), (2) a small group of matched SEER patients (SEER(M)), and (3) study participants (CUSIOS). The matched SEER group curves were created by randomly selecting several matched patients per study participant, with the number selected equalling the lowest number of matches of all the study participants in the given group (e.g., if the lowest number of matches was 4, then 4 SEER patients were included for each study participant). As the Kaplan-Meier curves were created as a visual representation of the data, no statistical analyses were applied. The complete bootstrapped analysis provides the best estimate for the confidence intervals.
Survival based on the type and frequency of naturopathic treatments received was conducted using a time varying covariate analysis and the log rank test. Univariable and multivariable models were constructed using the number of IVC, IV mistletoe, and hyperthermia treatments, number of ND visits, sex, age, histology, cancer stage, and hormone receptor status (breast cancer only) as covariates. Exact dates were used for ND visits, and time points of 3, 6, 9, 12, 18, 24, 30, and 36 months used for IVC, IV mistletoe, and hyperthermia treatments. Locoregional hyperthermia and whole-body hyperthermia were combined to increase power for this analysis. Results are presented showing the effect of the median number of treatments received.
Finally, some variables were analyzed using frequency distributions and descriptive statistics. Standard deviation was used as a method of dispersion for mean values. Inter-quartile range (IQR) or range was used as a method of dispersion for median values.
Data Availability Statement
Datasets used in this study are available by request only. Please contact Dugald Seely, dseely@thechi.ca, if you would like to request access to any datasets.
RESULTS
Participant Characteristics
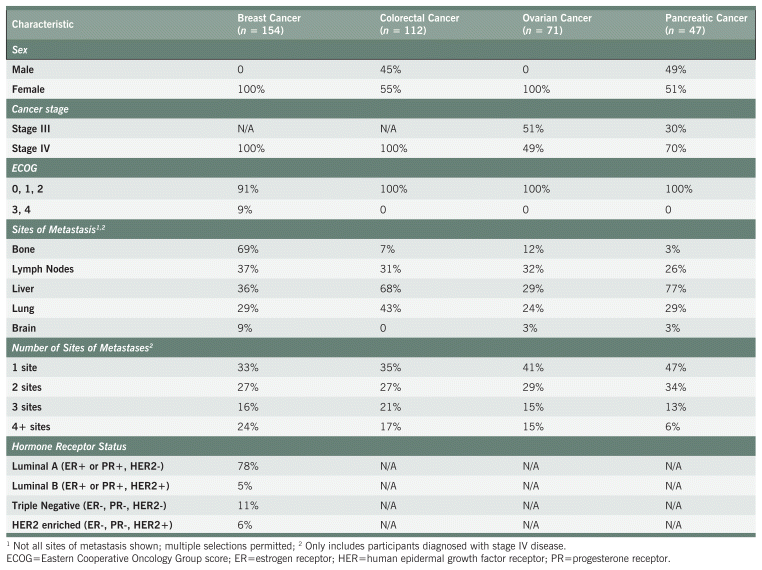
In total, 400 patients were enrolled. Figure 1 shows participant flow throughout the study and Table 1 summarizes baseline clinical status and demographic characteristics for all cancer types.
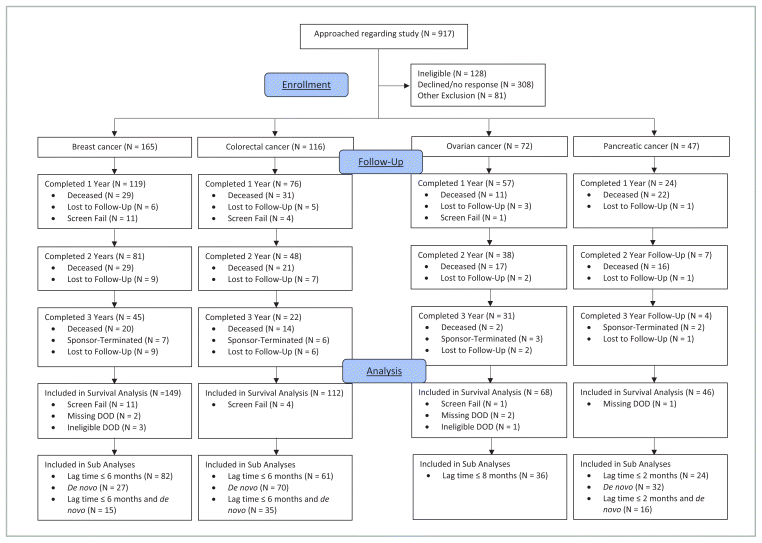
FIGURE 1 Participant Flow Diagram. Patients were only recorded as approached if an investigator believed they were eligible for the study and if the patient was interested in participating. Screen fail: patient was enrolled in the study but was later found to be ineligible; DOD: date of death.
TABLE 1 Clinical Status and Demographics
Breast Cancer
One hundred sixty-five participants had breast cancer. Eleven were found to be ineligible after enrolment and were not included in any analyses. The median time from participants’ diagnosis to their first ND visit was 5 months (IQR 2–14). Median time from the first ND visit to enrolment was 1 month (IQR 0–3). Mean age at diagnosis was 53.4 ± 12.0 years. 74% of participants had a ductal carcinoma; 16% had a lobular carcinoma. 83% were estrogen or progesterone receptor positive, and 11% were human epidermal growth factor receptor positive.
Colorectal Cancer
One hundred sixteen participants had colorectal cancer. Four were found to be ineligible after enrolment and were not included in any analyses. The median time from participants’ diagnosis to their first ND visit was 5 months (IQR 2–19). Median time from the first ND visit to enrolment was 1 month (IQR 0–3). Mean age at diagnosis was 55.8 ± 11.9 years. All participants had adenocarcinoma histology.
Ovarian Cancer
Seventy-two participants had ovarian cancer. One participant was found to be ineligible after enrolment and was not included in any analyses. The median time from participants’ diagnosis to their first ND visit was 7 months (IQR 2–26). Median time from the first ND visit to enrolment was 1 month (IQR 0–3). Mean age at diagnosis was 58.6 ± 10.6 years. 83% of participants had a serous carcinoma; 17% were classified as “other” histology. At the time of enrolment, 36 participants (51%) had stage III disease and 35 (49%) had stage IV. All participants with stage III disease were diagnosed de novo. Of those with stage IV disease, 26 (74%) were diagnosed de novo.
Pancreatic Cancer
Forty-seven participants had pancreatic cancer. The median time from participant diagnosis to their first ND visit was 1 month (IQR 1–7). Median time from the first ND visit to enrolment was 1 month (IQR 0–2). Mean age at diagnosis was 62.0 ± 11.6 years. 91% of participants had adenocarcinoma histology; 9% were classified as “other.” At the time of enrolment, 14 participants (30%) had stage III disease and 33 (70%) had stage IV. Of those with stage III disease, 13 (93%) were diagnosed de novo. Of those with stage IV disease, 19 (58%) were diagnosed de novo.
External Survival Comparison – SEER
Breast Cancer
Of the 165 participants with breast cancer, 149 were included in the primary survival analysis (see Figure 1 for exclusions). From SEER, there were 35,551 patients with stage IV breast cancer diagnosed in 2010 or later, 14,049 (40%) of whom matched with at least one study participant. Figure 2 shows survival results for SEER data compared with all 149 study participants. Survival favoured the SEER group but was not statistically significant (median HR 1.10, 95% CI 0.80–1.54). 894 patients were used to generate the matched SEER curve (least number of matches = 6).
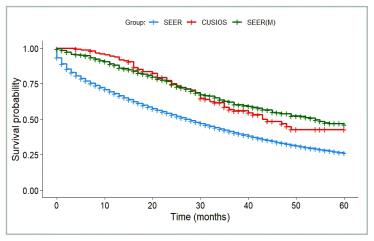
FIGURE 2 Breast Cancer Survival. SEER: All patients from SEER with stage IV breast cancer (N = 35,551). SEER(M): Patients from SEER who matched with at least one study participant (N = 894). CUSIOS: Study population (N = 149). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
In the subset of study participants who had a lag time between diagnosis and enrolment of 6 months or less (n = 82), survival favoured study participants but was not statistically significant (median HR 0.82, 95% CI 0.52–1.31). In the subset of study participants who were diagnosed with de novo stage IV disease (n = 27), survival favoured study participants but was not statistically significant (median HR 0.81, 95% CI 0.35–1.91). In the subset of study participants who were diagnosed with de novo stage IV disease and had a lag time between diagnosis and enrolment of 6 months or less (n = 15), survival favoured study participants but was not statistically significant (median HR 0.69, 95% CI 0.20–2.47). Kaplan Meier curves for each sub-analysis can be found in Appendix A, Figures A1–A3.
Colorectal Cancer
Of the 116 participants with colorectal cancer, 112 were included in the primary survival analysis (see Figure 1 for exclusions). From SEER, there were 71,735 patients with stage IV colon or rectal cancer diagnosed in 2010 or later, 22,156 (31%) of whom matched with at least one study participant. Figure 3 shows survival results for SEER data compared with all 112 participants. Survival did not favour either group (median HR 0.95, 95% CI 0.68–1.31). 2,688 patients were used to generate the matched SEER curve (least number of matches = 24).
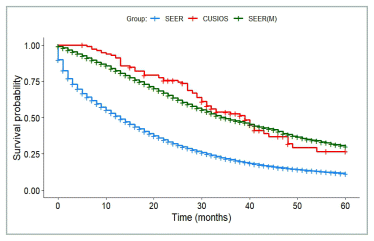
FIGURE 3 Colorectal Cancer Survival. SEER: All patients from SEER with stage IV colorectal cancer (N = 71,735). SEER(M): Patients from SEER who matched with at least one study participant (N = 2,688). CUSIOS: Study population (N = 112). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
In the subset of study participants whose lag time between diagnosis and enrolment was 6 months or less (n = 61), survival favoured study participants but was not statistically significant (median HR 0.64, 95% CI 0.40–1.04). In the subset of study participants who were diagnosed with de novo stage IV disease (n = 70), survival did not favour any group (median HR 0.99, 95% CI 0.66–1.49). In the subset of study participants who were diagnosed with de novo stage IV disease and had a lag time between diagnosis and enrolment of 6 months or less (n = 35), survival favoured study participants but was not statistically significant (median HR 0.63, 95% CI 0.32–1.21). Kaplan Meier curves for each sub-analysis can be found in Appendix A, Figures A4–A6.
Ovarian Cancer
Of the 72 participants with ovarian cancer, 68 were included in the primary survival analysis (see Figure 1 for exclusions). From SEER, there were 32,487 patients with stage III or IV ovarian cancer diagnosed from 2010 to 2019 or later, 7,101 (22%) of whom matched with at least one study participant. The bootstrapped survival analysis showed no significant differences between study participants and SEER (median HR 1.08, 95% CI 0.64–1.86). Figure 4 shows a Kaplan Meier curve for all study participants. 340 patients were used to generate the matched SEER curve (least number of matches = 5).
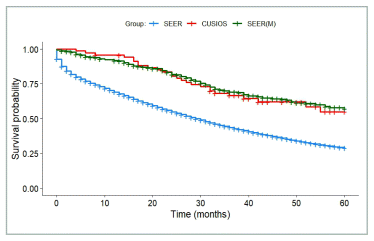
FIGURE 4 Ovarian Cancer Survival. SEER: All patients from SEER with stage III and IV ovarian cancer (N = 32,847). SEER(M): Patients from SEER who matched with at least one study participant (N = 340). CUSIOS: Study population (N = 68). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
In the subset of study participants who had a lag time between diagnosis and enrolment of 8 months or less (n = 36), the bootstrapped survival analysis showed no significant differences between study participants and SEER (median HR 0.98, 95% CI 0.47–2.11). Appendix A, Figure A7 shows the Kaplan Meier curve for this subset.
As per the study methods, ovarian cancer was omitted from any analyses involving de novo disease, given more than 90% of participants were diagnosed de novo.
Pancreatic Cancer
Of the 47 participants with pancreatic cancer, 46 were included in the primary survival analysis (see Figure 1 for exclusions). From SEER, there were 65,851 patients with stage III or IV pancreatic cancer diagnosed in 2010 or later, 15,075 (23%) of whom matched with at least one study participant. Figure 5 shows survival results for SEER data compared with all 46 study participants. Survival favoured study participants but was not statistically significant (median HR 0.76, 95% CI 0.51–1.13). 552 patients were used to generate the matched SEER curve (least number of matches = 12).
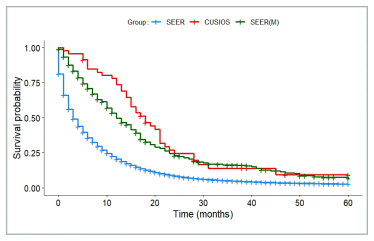
FIGURE 5 Pancreatic Cancer Survival. SEER: All patients from SEER with stage III or IV pancreatic cancer (N = 65,851). SEER(M): Patients from SEER who matched with at least one study participant (N = 552). CUSIOS: Study population (N = 46). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results
In the subset of study participants who had a lag time between diagnosis and enrolment of 2 months or less (n = 24), survival favoured study participants but was not statistically significant (median HR 0.62, 95% CI 0.29–1.14). In the subset of study participants who were diagnosed with de novo disease (n = 32), survival favoured study participants but was not statistically significant (median HR 0.79, 95% CI 0.49–1.29). In the subset of study participants who were diagnosed with de novo disease and had a lag time between diagnosis and enrollment of 2 months or less (n = 16), survival favoured study participants but was not statistically significant (median HR 0.70, 95% CI 0.24–1.51). Kaplan Meier curves for each sub-analysis can be found in Appendix A, Figures A8–A10.
Internal Survival Comparison – Treatment-Based
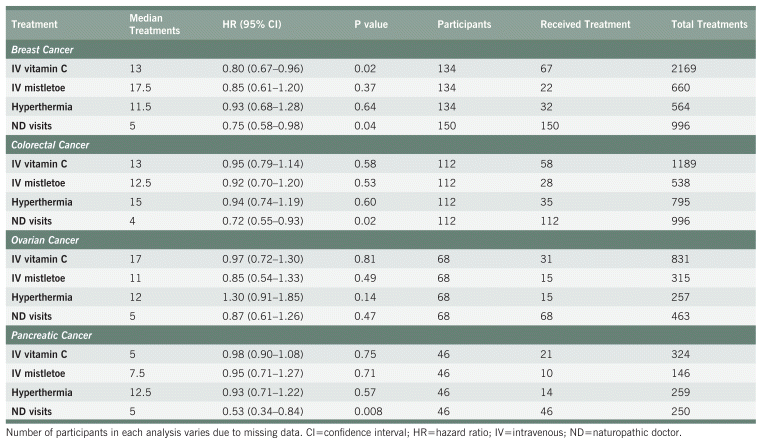
Table 2 shows the results from the treatment-based survival comparisons. Statistically significant improved survival odds were seen in participants with breast cancer who received more IVC treatments (HR 0.80, 95% CI 0.67–0.96, p = 0.02 for 13 additional treatments). Statistically significant improved survival odds were also seen in participants with breast, colorectal, and pancreatic cancer who saw their ND more often (breast: HR 0.75, 95% CI 0.58–0.98, p = 0.04 for 5 additional visits, colorectal: HR 0.72, 95% CI 0.55–0.93, p = 0.02 for 4 additional visits; pancreatic: HR 0.53, 95% CI 0.34–0.84, p = 0.008 for 5 additional visits). All results remained significant in the multivariate models. Intravenous mistletoe and hyperthermia treatments did not confer higher survival odds with increasing treatment numbers in either the univariate or multivariate models for any cancer type. A significant survival disadvantage was seen in the multivariate model for participants with ovarian cancer who received more hyperthermia treatments (HR 1.70, 95% CI 1.01–2.84, p = 0.05 for 12 additional treatments); however, this was not seen in the univariate model. Multivariate results are not shown in Table 2. Results from the multivariate analysis and HRs for a single additional treatment are presented in Appendix B.
TABLE 2 Survival Based on Naturopathic Treatments
Conventional Cancer Treatments
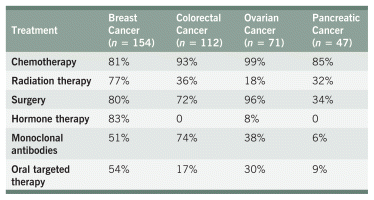
Table 3 shows the frequency of conventional cancer treatments received by participants. Almost all participants (99%) received some form of treatment either before or after they were enrolled. Four participants (1 breast cancer, 3 pancreatic cancer) did not receive any conventional cancer treatments.
TABLE 3 Conventional Cancer Treatments Received
DISCUSSION
In this multicentre North American observational study, we compared 5-year overall survival in patients with advanced and/or metastatic breast, colorectal, ovarian, or pancreatic cancer who consulted with an ND with SEER data. No significant differences were seen, including in multiple sub-group analyses of study participants who were diagnosed with de novo disease and who saw an ND shortly after their diagnosis. To our knowledge, this is the second study comparing the survival of patients with breast cancer receiving care from an ND with US registry data, and the first in patients with colorectal, ovarian, and pancreatic cancer. Researchers previously conducted a similar study assessing recurrence of breast cancer after primary treatment for earlier-stage disease, which showed no benefit in disease-free survival for patients receiving naturopathic care.20
Although no results from the SEER comparison were statistically significant, there are a few findings which are notable. Firstly, survival favoured study participants with pancreatic cancer with consistent hazard reductions across the main analysis and all sub-analyses. This is hypothesis-generating for future research, particularly given the small sample size for this cohort of patients. Secondly, across all 4 cancer types, participants with shorter intervals between diagnosis and study enrolment demonstrated more favourable survival times compared with the overall cohort. Furthermore, this sub-analysis showed non-significant hazard reductions in participants with breast, pancreatic, and colorectal cancer. For ovarian cancer, it was equivocal. This suggests patients seeking naturopathic care long after they were diagnosed may be different from those who seek care more quickly. For example, they may have done so due to disease progression or increased symptom burden, which may not be the case for the SEER patients with whom they were matched. Additionally, patients receiving care late in their disease trajectory may have had less opportunity for naturopathic therapies to have a clinically meaningful effect. These confounders could bias outcomes in favour of SEER. Alternatively, patients who seek naturopathic care soon after they are diagnosed may be biased in favour of a greater preference for naturopathic care and a higher socioeconomic status, given that they are willing to pay privately to see an ND right away. This could bias outcomes in favour of study participants. It is also important to note that we do not have data on adherence to ND recommendations or other important confounders, such as smoking status and adherence to guideline-appropriate conventional care, which could heavily influence outcomes. Although the hazard reductions for those with shorter lag times are hypothesis-generating, we cannot make any definitive conclusions with the available data.
For some cancer types, survival trends differ for those with recurrent metastatic disease versus those diagnosed de novo. This is important as we included both groups of patients in our study, but SEER does not track progression. Thus, results will inherently be biased. For metastatic breast cancer, research has shown that patients with recurrent disease have worse survival outcomes than patients who are diagnosed de novo.21 Given our population mostly had recurrent metastatic disease, this biases outcomes in favour of SEER. We saw more favourable outcomes in our de novo analysis for participants with breast cancer, which corroborates this bias. For patients with colorectal cancer, there are no reported differences between recurrent metastatic and de novo disease.22 For patients with pancreatic cancer, research shows more favourable outcomes for those with recurrent metastatic disease,23 which biases outcomes in favour of study participants. We did not see a difference between the main analysis and the de novo analysis for participants with pancreatic cancer, which suggests the bias may not have impacted this group.
We additionally compared survival among enrolled participants based on the number of naturopathic treatments received. Results indicated higher survival odds for participants receiving more ND visits if they had breast, colorectal, or pancreatic cancer. We also observed higher survival odds for participants receiving more IVC treatments if they had breast cancer. These results should be interpreted with caution, as the study was not specifically designed to evaluate these outcomes. Although we used a multivariate model to analyze the data, factors such as smoking status, socioeconomic status, and adherence to guideline-appropriate conventional care were once again not accounted for. The most noteworthy result is the positive correlation between number of ND visits and survival. A possible interpretation is that patients frequently seeing their ND may be more engaged in their treatment plan, and thus more likely to adhere to it. If this were true, it could suggest that those who more closely follow the recommendations of their NDs have improved survival outcomes compared with those who do not. While this is an interesting hypothesis, it relies on several assumptions and cannot be proven with the data available. Additionally, although the increase in survival with increasing IVC treatments in participants with breast cancer is encouraging, this effect was not seen in the other cancer types. There are currently no clinical trials evaluating IVC for survival outcomes in advanced breast cancer; however, there is supportive evidence for a survival advantage in metastatic pancreatic cancer24 and RAS-mutated colorectal cancer,25 which were not demonstrated herein. The number of hyperthermia treatments was negatively correlated with survival in the multivariate analysis of people with ovarian cancer; however, given these effects were not seen in the univariate model, it is likely not clinically significant. Finally, it is worth considering that people who choose to initiate IV therapy or hyperthermia may differ from those who do not. Those who choose these therapies may be doing so due to higher disease burden or progressive disease, which would negatively correlate with survival. Conversely, these individuals may have a higher socioeconomic status, which is often positively associated with survival. Because of this and the exploratory nature of the analysis, we cannot make any firm conclusions about the effect of naturopathic treatments on survival in our population.
Almost all participants received some form of conventional medical treatment either before or during the study. The frequency of use of these treatments in our population is comparable to, or higher than, population level data in the United States;26–28 however, there is one notable exception. The rate of endocrine therapy usage in study participants with breast cancer is lower than what is typically seen in the United States (68% versus 84%, respectively).27 These data are consistent with previous research that people with breast cancer receiving naturopathic care may be less likely to use endocrine therapy compared with those not receiving naturopathic care.29 This may reflect different treatment preferences of patients who seek ND care. Given the importance of endocrine therapy in management of breast cancer, this may negatively impact survival outcomes.30
Justification for Analysis
The SEER registry was chosen as our comparator for survival and only included patients who matched with a study participant using the variables described above. This matching process provided over 14,000 patient comparators. Traditionally, our cohort would be directly compared with all matched patients in SEER; however, this introduces bias, given that SEER data are population-based and do not control for certain clinical characteristics known amongst our cohort. Comparing a population with a small cohort is inherently biased, as covariates, such as clinical status and baseline prognostic factors, are much more impactful in the small cohort.31 To account for this bias, we used matched cohorts of equal size and bootstrapping to resample the data. The 1:1 comparison ensures covariates are equitably distributed, and the resampling increases the precision of our estimate of the hazard ratio and confidence interval. Using a matched population is also essential to control for confounding biases. Especially important was matching on lag time, whereby participants were only matched if their SEER counterpart survived at least as long as the lag time between diagnosis and enrolment in the study. This controls for immortal time bias. Immortal time is defined as a period where an event (mortality in this case) cannot occur. It is impossible for study participants to die during the time between diagnosis and enrolment; thus, it is important to compare them to registry patients who have survived at least as long. This is noticeable in our analysis, as both study participants and the population of matched SEER patients severely outperformed the global SEER population. This was almost certainly due to immortal time bias. Finally, the matched SEER Kaplan-Meier curves do not include all matched patients, rather, they include the lowest number of matches of all the study participants in the given group. Standardization to the least number of matches for each participant ensures that participants who could have a large number of matches do not skew the data. As previously mentioned, no statistical analyses were applied to these curves as they were solely intended to be a graphic representation of the data. Any statistical analysis would be inferior to what was generated using the matched bootstrap analysis.
The internal survival comparison was conducted using a time-varying covariate analysis. This type of analysis adjusts for the fact that covariates change during the follow-up period. Specifically, with regards to our study, there is a bias whereby the longer participants survive, the more likely they are to receive more treatments. This selection bias is accounted for and corrected in a time-varying covariate analysis. In our analysis, we compared participants who received at least one treatment with participants who received zero treatments, looking at the effect of the median number of treatments in each group. As an example, we saw a hazard ratio of 0.80 (20% reduction in risk of death) for participants receiving IVC in the breast cancer cohort. This means the hazard rate for a participant receiving 13 IVC treatments would be 0.8 times the hazard rate for a participant receiving zero treatments. The median number of treatments was used as a comparator; however, using the statistical model, the hazard ratio can be adjusted for any number of treatments while keeping the same P value.
Limitations
This study has several important limitations. The use of SEER as a comparator is a limitation for several reasons. Firstly, SEER does not collect data on cases of cancer progression and thus does not capture patients with recurrent metastatic disease, rather only de novo cancer staging. This was partially controlled in our sub-analyses for participants diagnosed with de novo disease; however, this was a relatively small proportion of our population. Practical constraints prevented us from only enrolling patients with de novo stage IV disease due to the low incidence of initial stage IV diagnoses, particularly with breast cancer.32 Secondly, there are inherent biases comparing population-level data with small cohorts as described above. This was at least partially controlled by our bootstrap analysis method. Thirdly, SEER does not gather data on Canadian patients; however, according to the American Cancer Society and Canadian Cancer Society, survival rates appear to be similar between Canada and the United States for all cancer types.3,33 SEER also does not capture important confounders, such as smoking status, socioeconomic status, and conventional treatment regimens. Finally, data on the usage of TCIM therapies are also not present in SEER, which may introduce a dilutional confounding bias.
Another limitation is the lack of exclusion criteria based on date of diagnosis. Each enrolled participant had a lag time from diagnosis to enrolment, which if uncontrolled creates an immortal time bias. We addressed this bias by only including SEER matches who survived at least as long as the lag time between diagnosis and enrolment in study participants. However, this method does not eliminate all bias that results from lag time. For example, there may be reasons why someone seeks ND care long after their diagnosis, such as progression or increased symptom burden, which may negatively impact their survival outcomes. Another consequence of this lag time is that participants may have received different conventional treatments or were at different stages of treatment at the time of enrolment, which decreases the internal validity of the study. This lag time created another challenge given that the primary outcome was 5-year overall survival. Ten participants (2.7%) had a lag time longer than 5 years, which meant their follow-up time on the study was not included. Additionally, some participants had lag times between 24 and 60 months, which meant their follow-up time on the study was truncated. While 5-year survival is a common measure and truncating at 5 years helps reduce some of the bias of long-term survivors, it introduces new bias in that participant follow-up while on-study (i.e., when participants were potentially receiving naturopathic care) was removed. We attempted to control for this bias by including a sub-analysis of participants with shorter lag times.
There is likely selection bias from a few different sources. Firstly, there was no mechanism in place to ensure participants were screened and enrolled consecutively. Secondly, it is well documented that individuals with cancer who seek complementary healthcare services have more comprehensive insurance coverage, are more likely to be non-smokers, and have higher education and income,34–36 all of which can affect clinical outcomes. Although these are general trends, we do not have specific data for our population.
Although some data were collected regarding conventional treatments received by study participants, the granularity of the data was not sufficient to assess whether participants adhered to standard oncologic care guidelines. Adherence to standard treatments would be expected to impact clinical outcomes.
Beyond limitations in study methodology, there are several other important considerations when interpreting these data. This study assessed 5-year survival as the primary outcome; however, likely not all participants sought naturopathic care to improve survival. Participants may have presented for side effect management or improving QOL but were included equally in the survival analysis. Data regarding adherence for most naturopathic therapies were not collected, and thus the degree to which participants followed the guidance provided by their ND is unknown. Furthermore, there are many participants who only saw their ND once. Therefore, it is important to recognize that the primary goal of the study was to assess whether at least one consultation with an ND, as delivered in real-world practice, affects survival. It does not assess the impact of consistent or optimal naturopathic care. While this could be considered a limitation, and valuable information can be gathered from assessing compliance, real-world research offers valuable insights into the typical outcomes expected in clinical practice.
The above limitations are difficult to address in an observational study. Given the challenges, it may not be appropriate to assess survival benefits by comparing a small cohort with registry data. It is clear from our analysis that using a matched population, especially the control of immortal time bias, is required at minimum. Additionally, enrolling patients shortly after diagnosis and limiting enrollment to those diagnosed de novo with the cancer stage under investigation would further mitigate potential bias. However, the lack of data regarding conventional treatment regimens and use of confounding treatments in SEER severely decreases internal validity, and selection bias will remain a concern. Ultimately, a clinical trial, whereby patients would be recruited in a hospital setting who are newly diagnosed and naïve to naturopathic care, would be the optimal way around these challenges. A trial of this nature is being conducted by a group in Ottawa, Canada, in which patients with newly diagnosed lung, gastric, or esophageal cancer are randomized to receive standard care plus naturopathic care or standard care alone.37 However, studies of this magnitude are complex, costly, and suffer from reductions in external validity when using standardized treatment protocols.
Strengths
There are a few strengths worth noting. This is the first study to evaluate survival outcomes for patients with advanced cancer receiving naturopathic care, and thus the methods and findings are novel. Additionally, the multicentre and international recruitment increases the generalizability of our findings. The prospective enrolment reduces selection bias. The statistical methods that were used for both the bootstrapped and time-varying covariate analysis helped reduce known biases with observational research. Lastly, the sub-analyses provide insights into the impact of important characteristics in our population and may be hypothesis-generating for future research.
CONCLUSION
This study demonstrated no survival advantages for patients with advanced breast, colorectal, pancreatic, or ovarian cancer who consulted with a naturopathic doctor compared with US registry data. Despite the lack of improvements, we cannot conclude that naturopathic medicine is ineffective for survival in this population due to the many biases and limitations in the study design which cannot be adequately controlled. This study is the first large-scale attempt to evaluate how consulting with a naturopathic doctor impacts cancer outcomes in a real-world setting. Further research in a more controlled setting, such as a clinical trial, is needed to clarify the role of naturopathic medicine for survival in people with advanced cancer.
AUTHOR AFFILIATIONS
1Bastyr University Research Institute, Washington, USA;
2Patterson Institute for Integrative Oncology Research, Ontario, Canada;
3The Centre for Health Innovation, Ontario, Canada;
4Integrated Health Clinic Cancer Care Centre, British Columbia, Canada;
5Marsden Centre for Excellence in Integrative Medicine, Ontario, Canada;
6Health Source Integrative Medical Centre, Ontario, Canada;
7Naturopathic Specialists, LLC. Arizona, USA;
8Salish Cancer Centre, Washington, USA;
9Hawaii Integrative Oncology – Lokahi Health Centre, Hawaii, USA;
10Tree of Health Integrative Medicine, Washington, USA;
11Ottawa Hospital Research Institute, Ontario, Canada
ACKNOWLEDGEMENTS
Not applicable
CONFLICTS OF INTEREST DISCLOSURE
We have read and understood the CAND Journal’s policy on conflicts of interest and declare that we have none.
FUNDING
This study was supported by the Lotte and John Hecht Memorial Foundation. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
REFERENCES
1. Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229–263. https://doi.org/10.3322/caac.21834
Crossref PubMed
2. Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA Cancer J Clin. 2025;75(1):10–45. https://doi.org/10.3322/caac.21871
Crossref PubMed PMC
3. Canadian Cancer Society. Canadian Cancer Statistics. 2023; https://cancer.ca/en/research/cancer-statistics. Date Accessed: March 11th, 2025
4. Horneber M, Bueschel G, Dennert G, Less D, Ritter E, Zwahlen M. How many cancer patients use complementary and alternative medicine: a systematic review and meta-analysis. Integr Cancer Ther. 2012;11(3):187–203. https://doi.org/10.1177/1534735411423920
Crossref
5. Wortmann JK, Bremer A, Eich HT, et al. Use of complementary and alternative medicine by patients with cancer: a cross-sectional study at different points of cancer care. Med Oncol. 2016;33(7):78–78. https://doi.org/10.1007/s12032-016-0790-4
Crossref PubMed
6. Judson PL, Abdallah R, Xiong Y, Ebbert J, Lancaster JM. Complementary and alternative medicine use in individuals presenting for care at a comprehensive cancer center. Integr Cancer Ther. 2016;16(1):96–103. https://doi.org/10.1177/1534735416660384
Crossref PubMed PMC
7. Steel A, Foley H, Bradley R, et al. Overview of international naturopathic practice and patient characteristics: results from a cross-sectional study in 14 countries. BMC Complement Med Ther. 2020;20(1):59. https://doi.org/10.1186/s12906-020-2851-7
Crossref PubMed PMC
8. Steel A, Redmond R, Schloss J, et al. International prevalence of consultation with a naturopathic practitioner: a systematic review and meta-analysis. BMJ Open. 2022;12(7):e056075. https://doi.org/10.1136/bmjopen-2021-056075
Crossref PubMed PMC
9. Snider P, Zeff J. Unifying principles of naturopathic medicine origins and definitions. Integr Med (Encinitas). 2019;18(4):36–39.
10. Greenlee H, DuPont-Reyes MJ, Balneaves LG, et al. Clinical practice guidelines on the evidence-based use of integrative therapies during and after breast cancer treatment. CA Cancer J Clin. 2017;67(3):194–232. https://doi.org/10.3322/caac.21397
Crossref PubMed PMC
11. Eliza WLY, Fai CK, Chung LP. Efficacy of Yun Zhi (Coriolus versicolor) on survival in cancer patients: systematic review and meta-analysis. Recent Pat Inflamm Allergy Drug Discov. 2012;6(1):78–87.
Crossref
12. Ostermann T, Appelbaum S, Poier D, Boehm K, Raak C, Büssing A. A systematic review and meta-analysis on the survival of cancer patients treated with a fermented Viscum album L. extract (Iscador): an update of findings. Complement Med Res. 2020;27(4):260–271. Eine Systematische Übersicht und Meta-Analyse zum Überleben von Krebspatienten, die mit einem Fermentiertem Viscum-Album-L.-Extrakt behandelt wurden: Ein Ergebnis-Update. https://doi.org/10.1159/000505202
PubMed
13. Friedenreich CM, Stone CR, Cheung WY, Hayes SC. Physical activity and mortality in cancer survivors: a systematic review and meta-analysis. JNCI Cancer Spectr. 2020;4(1):pkz080. https://doi.org/10.1093/jncics/pkz080
Crossref PubMed PMC
14. Morze J, Danielewicz A, Przybyłowicz K, Zeng H, Hoffmann G, Schwingshackl L. An updated systematic review and meta-analysis on adherence to Mediterranean diet and risk of cancer. Eur J Nutr. 2021;60(3):1561–1586. https://doi.org/10.1007/s00394-020-02346-6
Crossref PMC
15. Chen G, Leary S, Niu J, Perry R, Papadaki A. The role of the Mediterranean diet in breast cancer survivorship: a systematic review and meta-analysis of observational studies and randomised controlled trials. Nutrients. 2023;15(9):2099. https://doi.org/10.3390/nu15092099
Crossref PubMed PMC
16. McCulloch M, Broffman M, van der Laan M, et al. Colon cancer survival with herbal medicine and vitamins combined with standard therapy in a whole-systems approach: ten-year follow-up data analyzed with marginal structural models and propensity score methods. Integr Cancer Ther. 2011;10(3):240–259. https://doi.org/10.1177/1534735411406539
Crossref PubMed PMC
17. McCulloch M, Broffman M, van der Laan M, et al. Lung cancer survival with herbal medicine and vitamins in a whole-systems approach: ten-year follow-up data analyzed with marginal structural models and propensity score methods. Integr Cancer Ther. 2011;10(3):260–279. https://doi.org/10.1177/1534735411406439
Crossref PubMed
18. Seely D, Legacy M, Conte E, et al. A comprehensive description of naturopathic care for advanced cancers: outcomes from the Canadian/US integrative oncology study. CAND Journal. 2025;32(2):21–40. https://doi.org/10.54434/candj.207
19. SEER. SEER Program Coding and Staging Manual. 2023.
20. Standish LJ, Sweet E, Kim E, et al. Recurrence of breast cancer after primary treatment: a matched comparison study of disease-free survival in women who do and do not receive adjunctive naturopathic oncology care. Integr Cancer Thers. 2021;20:15347354211058404. https://doi.org/10.1177/15347354211058404
Crossref
21. File DM, Pascual T, Deal AM, et al. Clinical subtype, treatment response, and survival in de novo and recurrent metastatic breast cancer. Breast Cancer Res Treat. 2022;196(1):153–162. https://doi.org/10.1007/s10549-022-06700-6
Crossref PubMed PMC
22. Hassett MJ, Uno H, Cronin AM, Carroll NM, Hornbrook MC, Ritzwoller DP. Comparing survival after recurrent vs de novo stage IV advanced breast, lung, and colorectal cancer. JNCI Cancer Spectr. 2018;2(2):pky024. https://doi.org/10.1093/jncics/pky024
Crossref PubMed PMC
23. Miotke L, Nevala-Plagemann C, Ying J, Florou V, Haaland B, Garrido-Laguna I. Treatment outcomes in recurrent versus de novo metastatic pancreatic adenocarcinoma: a real world study. BMC Cancer. 2022;22(1):1054. https://doi.org/10.1186/s12885-022-10130-4
Crossref PubMed PMC
24. Bodeker KL, Smith BJ, Berg DJ, et al. A randomized trial of pharmacological ascorbate, gemcitabine, and nab-paclitaxel for metastatic pancreatic cancer. Redox Biology. 2024;77:103375. https://doi.org/10.1016/j.redox.2024.103375
Crossref PubMed PMC
25. Wang F, He MM, Xiao J, et al. A randomized, open-label, multicenter, phase 3 study of high-dose vitamin C plus FOLFOX ± bevacizumab versus FOLFOX ± bevacizumab in unresectable untreated metastatic colorectal cancer. Clin Cancer Res. 2022;28(19):4232–4239. https://doi.org/10.1158/1078-0432.CCR-22-0655
Crossref PubMed PMC
26. Giaquinto AN, Sung H, Miller KD, et al. Breast cancer statistics, 2022. CA Cancer J Clin. 2022;72(6):524–541. https://doi.org/10.3322/caac.21754
Crossref PubMed
27. Miller KD, Nogueira L, Devasia T, et al. Cancer treatment and survivorship statistics, 2022. CA Cancer J Clin. 2022;72(5):409–436. https://doi.org/10.3322/caac.21731
Crossref PubMed
28. Marth C, Abreu MH, Andersen KK, et al. Real-life data on treatment and outcomes in advanced ovarian cancer: an observational, multinational cohort study (RESPONSE trial). Cancer. 2022;128(16):3080–3089. https://doi.org/10.1002/cncr.34350
Crossref PubMed PMC
29. Standish LJ, Dowd F, Sweet E, Dale L, Andersen MR. Do women with breast cancer who choose adjunctive integrative oncology care receive different standard oncologic treatment? Integr Cancer Ther. 2018;17(3):874–884. https://doi.org/10.1177/1534735418769007
Crossref PubMed PMC
30. Manohar PM, Davidson NE. Updates in endocrine therapy for metastatic breast cancer. Cancer Biol Med. 2021;19(2):202–12. https://doi.org/10.20892/j.issn.2095-3941.2021.0255
PubMed PMC
31. Lord SJ, Bahlmann K, O’Connell DL, et al. De novo and recurrent metastatic breast cancer—a systematic review of population-level changes in survival since 1995. EClinicalMedicine. 2022;44:101282. https://doi.org/10.1016/j.eclinm.2022.101282
Crossref
32. Daily K, Douglas E, Romitti PA, Thomas A. Epidemiology of de novo metastatic breast cancer. Clin Breast Cancer. 2021;21(4):302–308. https://doi.org/10.1016/j.clbc.2021.01.017
Crossref PubMed
33. American Cancer Society. Cancer facts and statistics. Accessed 2025-03-10, https://www.cancer.org/research/cancer-facts-statistics.html
34. Wanchai A, Armer JM, Stewart BR. Complementary and alternative medicine use among women with breast cancer: a systematic review. Clin J Oncol Nurs. 2010;14(4):E45–55. https://doi.org/10.1188/10.Cjon.E45-e55
Crossref PubMed
35. Helpman L, Ferguson SE, Mackean M, et al. Complementary and alternative medicine use among women receiving chemotherapy for ovarian cancer in 2 patient populations. Int J Gynecol Cancer. 2011;21(3):587–93. https://doi.org/10.1097/IGC.0b013e31820fa24a
Crossref PubMed
36. Tough SC, Johnston DW, Verhoef MJ, Arthur K, Bryant H. Complementary and alternative medicine use among colorectal cancer patients in Alberta, Canada. Altern Ther Health Med. 2002;8(2):54–56, 58–60, 62–64.
PubMed
37. Seely D, Ennis JE, McDonell E, et al. Intervention development process for a pragmatic randomized controlled trial: the thoracic peri-operative integrative surgical care evaluation trial. J Altern Complement Med. 2019;25(S1):S112–S123. https://doi.org/10.1089/acm.2018.0419
Crossref PubMed
APPENDIX A: SEER SURVIVAL COMPARISON
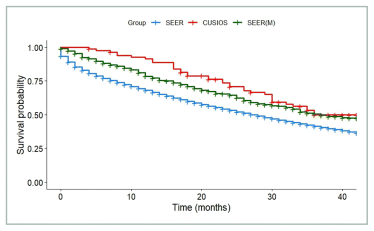
FIGURE A1 Breast cancer: survival of participants with lag time ≤ 6 months compared with SEER registry. SEER: All patients from SEER with Stage IV breast cancer (N = 35,551). SEER(M): Patients from SEER who matched with at least one study participant (N = 902). CUSIOS: Study population (N = 82). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
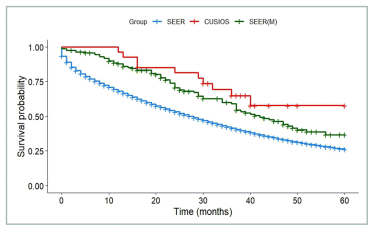
FIGURE A2 Breast cancer: survival of participants with de novo disease compared with SEER registry. SEER: All patients from SEER with Stage IV breast cancer (N = 35,551). SEER(M): Patients from SEER who matched with at least one study participant (N = 162). CUSIOS: Study population (N = 27). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
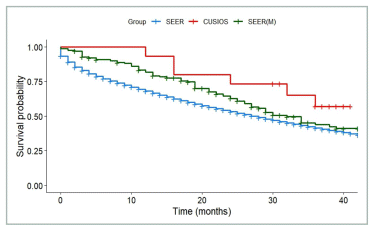
FIGURE A3 Breast cancer: survival of participants with de novo disease and lag time ≤ 6 months compared with SEER registry. SEER: All patients from SEER with Stage IV breast cancer (N = 35,551). SEER(M): Patients from SEER who matched with at least one study participant (N = 165). CUSIOS: Study population (N = 15). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
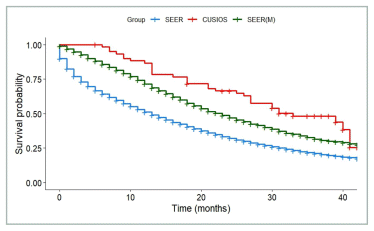
FIGURE A4 Colorectal cancer: survival of participants with lag time ≤ 6 months compared with SEER registry. SEER: All patients from SEER with Stage IV colorectal cancer (N = 71,735). SEER(M): Patients from SEER who matched with at least one study participant (N = 6,466). CUSIOS: Study population (N = 61). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
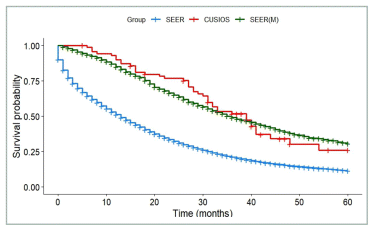
FIGURE A5 Colorectal cancer: survival of participants with de novo disease compared with SEER registry. SEER: All patients from SEER with Stage IV colorectal cancer (N = 71,735). SEER(M): Patients from SEER who matched with at least one study participant (N = 3,640). CUSIOS: Study population (N = 70). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
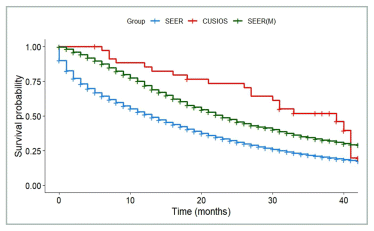
FIGURE A6 Colorectal cancer: survival of participants with de novo disease and lag time ≤ 6 months compared with SEER registry. SEER: All patients from SEER with Stage IV colorectal cancer (N = 71,735). SEER(M): Patients from SEER who matched with at least one study participant (N = 3,710). CUSIOS: Study population (N = 35). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
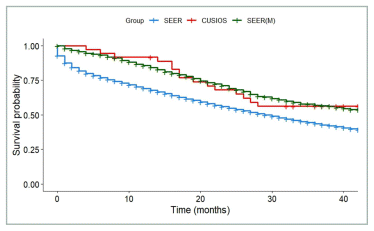
FIGURE A7 Ovarian cancer: survival of participants with lag time ≤ 8 months compared with SEER registry. SEER: All patients from SEER with Stage III or IV ovarian cancer (N = 32,847). SEER(M): Patients from SEER who matched with at least one study participant (N = 1,476). CUSIOS: Study population (N = 36). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
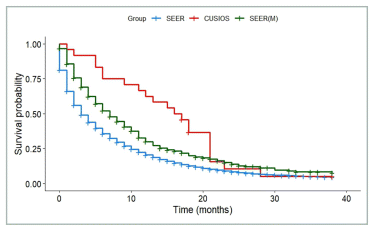
FIGURE A8 Pancreatic cancer: survival of participants with lag time ≤ 2 months compared with SEER registry. SEER: All patients from SEER with stage III or IV pancreatic cancer (N = 65,851). SEER(M): Patients from SEER who matched with at least one study participant (N = 624). CUSIOS: Study population (N = 24). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
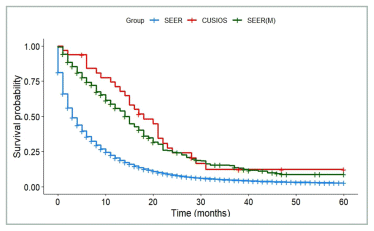
FIGURE A9 Pancreatic cancer: survival of participants with de novo disease compared with SEER registry. SEER: All patients from SEER with stage III or IV pancreatic cancer (N = 65,851). SEER(M): Patients from SEER who matched with at least one study participant (N = 384). CUSIOS: Study population (N = 32). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
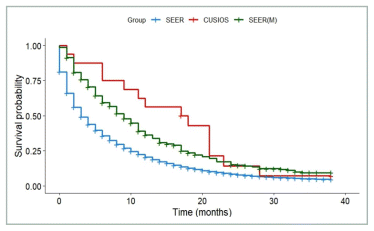
FIGURE A10 Pancreatic cancer: survival of participants with de novo disease and lag time ≤ 2 months compared with SEER registry. SEER: All patients from SEER with stage III or IV pancreatic cancer (N = 65,851). SEER(M): Patients from SEER who matched with at least one study participant (N = 416). CUSIOS: Study population (N = 16). This curve was created as a graphic representation to aid viewers in visualizing the data. Statistical analyses were only applied to the main bootstrapped analysis. SEER: Surveillance Epidemiology and End Results (registry data); CUSIOS: Canadian/US Integrative Oncology Study (study population).
APPENDIX B: INTERNAL SURVIVAL COMPARISON
TABLE A1 Survival Among Study Participants with Breast Cancer
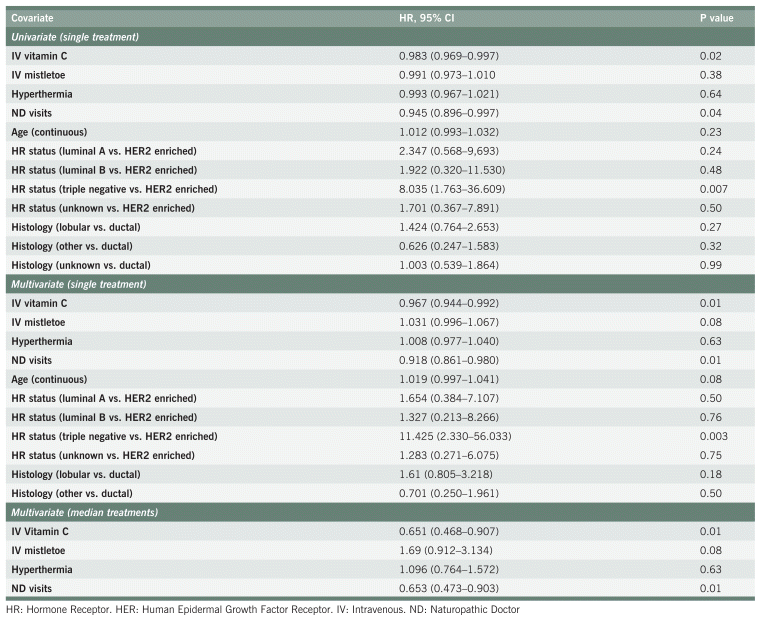
TABLE A2 Survival Among Study Participants with Colorectal Cancer
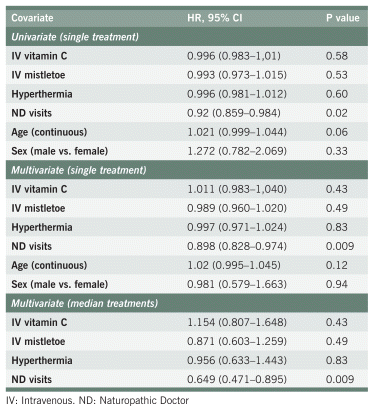
TABLE A3 Survival Among Study Participants with Ovarian Cancer
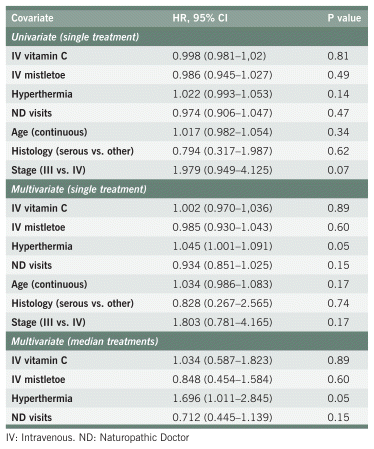
TABLE A4 Survival Among Study Participants with Pancreatic Cancer
Correspondence to: Leanna J. Standish, 2825 Eastlake Avenue East #115, Seattle, WA 98102, United States. E-mail: lstandish@aimsinstitute.net
To cite: Standish LJ, Sweet E, Chiang PF. Naturopathic oncology for advanced cancers: Survival outcomes from the Canadian/US integrative oncology study. CAND Journal. 2025;32(2):4-20. https://doi.org/10.54434/candj.206
Received: 13 March 2025; Accepted: 28 April 2025; Published: 19 June 2025
© Author(s) 2025. This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution-NonCommercial (CC BY-NC 4.0) license. See https://creativecommons.org/licenses/by-nc/4.0/. For commercial re-use, please contact candj@cand.ca.
Published by the Canadian Association of Naturopathic Doctors.
CAND Journal | Volume 32, No. 2, June 2025
(Return to Top)